Highlights
- Invion has made further significant progress in developing the PhotosoftTM technology in the September quarter.
- In a recent in vitro study, Invion's research partner Peter Mac demonstrated the effectiveness of INV043 against anal cancers.
- Invion achieved successful results from in vitro studies of PhotosoftTM on Zika and SARS-CoV-2 – delta and omicron variants.
The September quarter of 2022 was a busy period for Invion Limited (ASX:IVX), which reported significant results from its research & development (R&D) activities. The company's major focus remained on advancing the development of its PhotosoftTM technology to treat different types of cancer and infectious diseases.
The company is undertaking its R&D activities in collaboration with international institutes including the Peter MacCallum Cancer Centre and the Hudson Institute of Medical Research.
INV043 is effective against anal cancer
INV043 is Invion's lead API for the development of next generation of PDT cancer treatments.
The company’s research partner Peter Mac demonstrated the effectiveness of INV043 against anal cancers in a recent in vitro study. The compound was found to be effective against six squamous cell carcinoma (SCC) cell lines representing the full range of anal cancers.
The results of this study are in line with the positive outcomes achieved by the Hudson Institute on other types of cancers, including triple-negative breast cancer.
Invion's partners are commencing further work on INV043, which is expected to pave the way for human clinical trials. The company anticipates starting at least one of these trials in the first half of CY2023.
PhotosoftTM shows antiviral activity against Zika virus
The quarter saw the release of the first test results using PhotosoftTM on viruses. In in vitro studies conducted by Viroclinics-DDL and Virology Research Services Ltd (VRS), selected PhotosoftTM compounds were found to be effective against the Zika virus.
The study also found these compounds boasting a higher Therapeutic Index than the control Monensin.
Monensin is an antibiotic found to be effective against Zika in in vitro laboratory tests. However, due to its in vivo toxicity, Monensin cannot be used in humans.
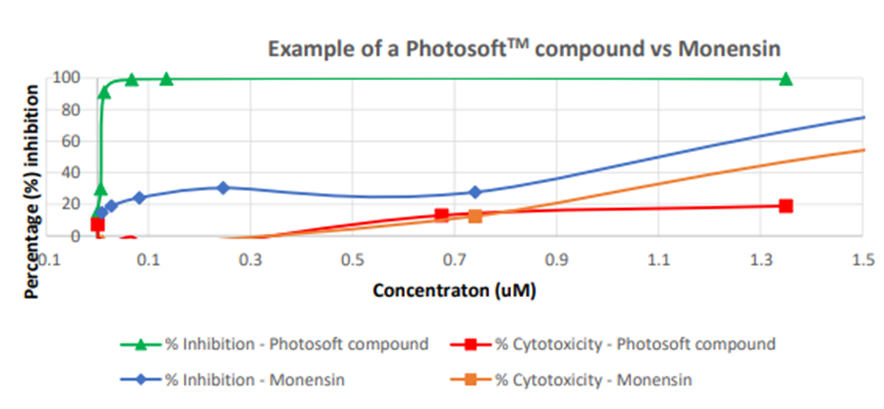
Image source: Company update
PhotosoftTM displays antiviral activity against COVID-19
In another set of a study conducted by Viroclinics and VRS, nine out of the ten PhotosoftTM compounds exhibited antiviral activity against the Delta and Omicron BA.1 variants of SARS-CoV-2.
The control used in this study was Remdesivir, a broad-spectrum antiviral medication that has been approved for emergency use in different nations to treat COVID-19.
Five of the tested compounds demonstrated no cytotoxicity, whereas the other five exhibited light-dependent cytotoxicity.
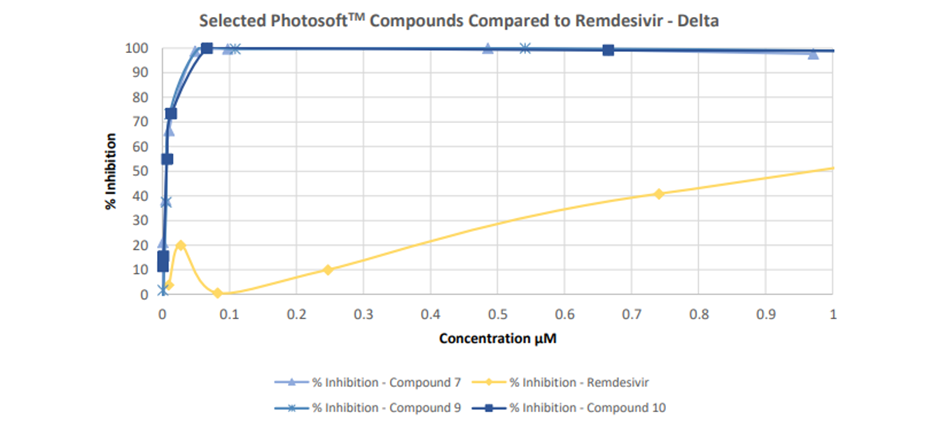 Image source: Company update
Image source: Company update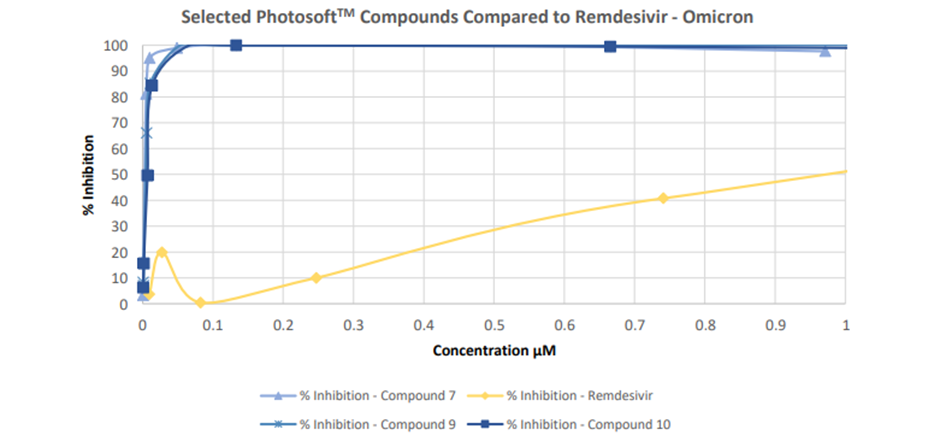
Image source: Company update
Financial update
Invion ended the quarter with a cash balance of AU$7.8 million and no debt. The company has a funding agreement with RMW Cho Group.
The company says that it is well-founded to pursue its near-and medium-term development agenda.
Invion shares traded at AU$0.010 with an upsurge of 5% on 02 November 2022.


_07_12_2023_11_05_52_936360.jpg)




