To increase the bioavailability of administered medications, Australian-headquartered SUDA Pharmaceuticals Ltd (ASX: SUD) is reformulating medications into an oral spray. The delivery of medications through the oral mucosa increases the amount of the drug absorbed by the body, thereby improving the drug effectiveness. SUDA has a strong market interest in Cannabinoids (CBD), and its OroMist® oro-mucosal spray technology provides a good platform for CBD products.
In its latest update on the ASX, the company notified that it supports the Australian Governmentâs initiative regarding the medical marijuana industry. Health Minister, Mr Greg Hunt has recently informed that the Government will foster the national medical marijuana industry by giving priority for medical marijuana licences to projects that have been granted Major Project Status (MPS). The Major Project Status is overseen by the Department of Industry in Australia.
An Insight into Department of Healthâs Announcement
On 2nd August 2019, the Australian Governmentâs Department of Health released the media announcement to prioritise medicinal cannabis projects with MPS status. The Major Project Status is granted to those projects in Australia that are expected to provide a valuable contribution in the areas of jobs, industry development and exports, predominantly in regional and rural areas.
The Australian Government will work towards a single licence model and will be seeking ways to remove impediments that congested administration and development of the medicinal cannabis industry. According to Mr Hunt, this cooperative mechanism between the Department of Industry and the Department of Health showcases the Governmentâs commitment to supporting those suffering from chronic or terminal illnesses and giving a boost to local industry.
As per the announcement, the Governmentâs move will lift Australiaâs medicinal cannabis industry, ultimately driving the economic growth. Medical marijuana licences with MPS status will be granted from 1 September 2019 through the Department of Healthâs ODC (Office of Drug Control).
The Office of Drug Control, that governs the licenses for the manufacturing, cultivation and the delivery of medicinal cannabis, has so far issued 69 licenses in Australia. These licences include 16 for cultivation for research, 24 for the cultivation of cannabis for medicinal use, and 23 for production of medicinal cannabis products.
The Department of Healthâs media release mentioned that more than 10k prescriptions for medicinal cannabis products have been written in Australia as on 30th June 2019. Also, more than 7000 have been permitted in Australia to obtain a medicinal cannabis product via the Authorised Prescriber and Special Access Schemes.
SUDAâs Licenses in Connection with Cannabinoid Projects
The oro-mucosal drug delivery company has been developing its infrastructure to conduct research into Medical Marijuana. SUDA possesses all the essential licenses required to conduct research and development on cannabis-based products.
So far, the company has received following licences related to its cannabinoid projects from either the Government of Western Australia, the Australia Therapeutic Goods Administration or the ODC:
- License to export and import cannabinoid and cannabis products,
- License to buy cannabinoids and cannabis for the research and education purpose,
- Good Manufacturing Practice Licence that permits the manufacturing of initial stage clinical samples (up to Phase 1) and release for delivery,
- Wholesale Warehousing licence that permits the delivery of medicines (including cannabis) for clinical trialsâ use, and
- Permit to buy and own tetrahydrocannabinols, cannabidiol, and cannabinoids.
SUDAâs Progress in Medical Marijuana Industry
SUDA has made substantial progress in the field of the medical marijuana industry in the last year. The company has entered into agreements with two pharmaceutical companies to assess the feasibility of developing medical-grade cannabis using its Hydrotrope and OroMist® platforms.
Let us take a look at the agreements signed by the company in the last twelve months:
Agreement with Cann Pharmaceutical Australia Ltd
In June 2019, the company signed a binding term sheet agreement with Cann Pharmaceutical Australia Ltd or CPA for an exclusive licence to develop and deliver an oral spray of pharmaceutical-class cannabinoid derivatives. CPA is focussing on the formulation of a novel oral spray of pharmaceutical-class cannabinoid derivatives for the treatment of melanoma, drug-resistant epilepsy and motion sickness.
The company informed that CPA would fully fund the development of the product and look into the global regulatory approvals for the product. As per the company, the cost of the initial development phase and permeability testing was anticipated to be ~$184k. SUDA and CPA will determine the other development costs at a later stage.

Agreement with Zelda Therapeutics Ltd
Under the one-year workplan, SUDA was to apply its proprietary oro-mucosal spray technology, OroMist®, to Zeldaâs pharmaceutical-class cannabinoid derivatives, for assessment by Zelda.
The agreement entitled Zelda to pay an option fee of $200,000 to SUDA that included an upfront of $100k and a further $100k in downstream milestone payments. A 24-month option was provided to Zelda under which Zelda had an exclusive right to stretch the Agreement and to sign a licensing and global development agreement for the oral spray formulations of SUDA.
Besides cannabinoid products, there are many other products in the SUDAâs product pipeline that are in the development phase. Those products include oral sprays for the treatment of chemotherapy-induced nausea and vomiting, PAH, migraine headache, cancer, epileptic seizures, erectile dysfunction and pre-procedural anxiety. SUDA also holds exclusive license agreements with different companies for its US FDA approved product, ZolpiMistâ¢, an oral spray formulation targeting insomnia.
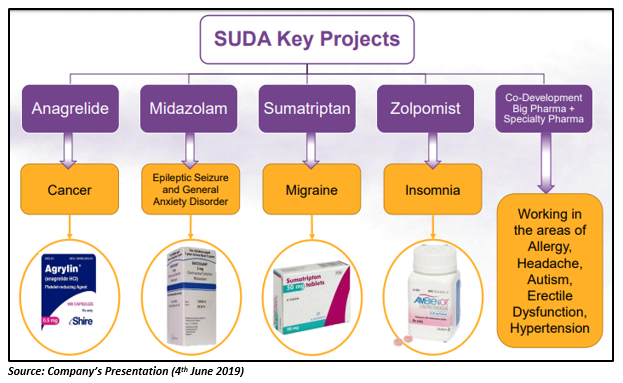
By now, the company has made speedy progress with its product pipeline in the evolution of drug delivery technology. The company is utilizing its extensive knowledge of proven techniques to address the core issues of drug efficacy.
Stock Performance: The companyâs stock is currently trading at AUD 0.004 on 9th August 2019 (11:38 AM AEST). SUDA has delivered an enormous return of 28.57 per cent in the last one month.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. The above article is sponsored but NOT a solicitation or recommendation to buy, sell or hold the stock of the company (or companies) under discussion. We are neither licensed nor qualified to provide investment advice through this platform.







