Highlights
- Invion is going strong with research work focused on cancer and infectious diseases.
- The lead drug candidate INV043 has demonstrated promising results in studies covering triple negative breast cancer and anal cancers.
- INV043 has also shown encouraging results against infectious diseases such as COVID-19, Zika virus and Dengue.
- With primary focus on cancer, the company expects to commence Phase I clinical trials in 2023.
Australia-based life sciences company Invion Limited (ASX:IVX) is gearing up for what could be a very productive and exciting 2023.
It’s making major headways to transform Photodynamic Therapy (PDT) for the treatment of cancer, atherosclerosis, and infectious diseases. The company is focused on the research & development of Photosoft™ technology as novel, next-generation PDT.
Invion is progressing PhotosoftTM technology related cancer research programs in collaboration with prominent global medical research institutes including the Hudson Institute of Medical Research and the Peter MacCallum Cancer Centre. Meanwhile, the company has partnered with the Australian Centre for Antimicrobial Resistance Ecology and Viroclinics for research in infectious diseases.
Key achievements in cancer research
Invion is primarily focused on cancer research. The company says that it is gearing up to commence Phase 1 clinical trials in 2023.
Its lead drug candidate for cancer is INV043, providing a pathway to participate in the huge oncology market opportunity worth US$271 billion (as per GMI, IVX update).
During in vivo tests, INV043 has demonstrated complete regression of triple negative breast cancer (TNBC) in a proof-of-concept (PoC) primary tumour pilot study. It also induced protective immunity as there was no disease recurrence upon rechallenging with TNBC implant.
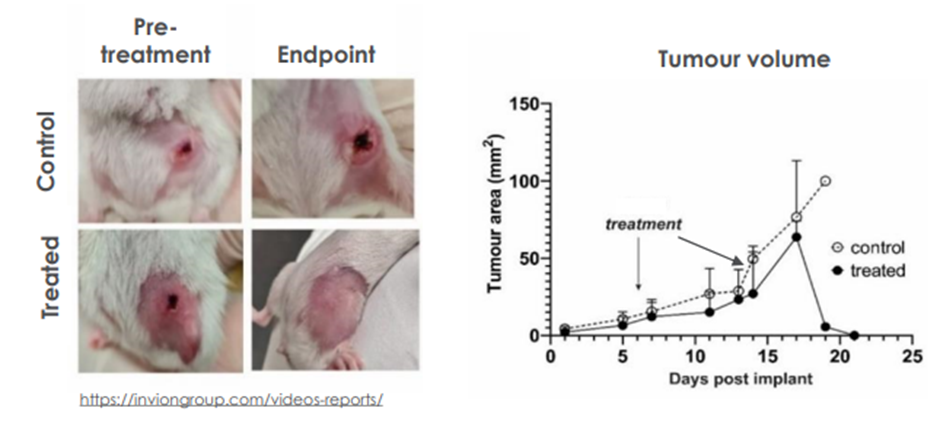
Image source: IVX update
Image description: PDT using INV043 results in complete regression of established tumours
INV043 is also being studied against anal cancer. Under an in vitro study, the compound showed its effectiveness against six squamous cell carcinoma (SCC) cell lines representing the full range of anal cancers.
Moreover, INV043 has exhibited good synergy with the blockbuster Immune Checkpoint Inhibitors (ICI) drugs. A Hudson Institute-conducted PoC study assessed the effect of combining INV043 with ICI targeting PD-1. The combination therapy significantly improved tumour growth restriction (~65%). There was clear tumour regression despite the use of sub-optimal INV043 treatment protocol.
ICI is a major part of the immunotherapy space; however, treatments are generally effective only in a small proportion of patients.
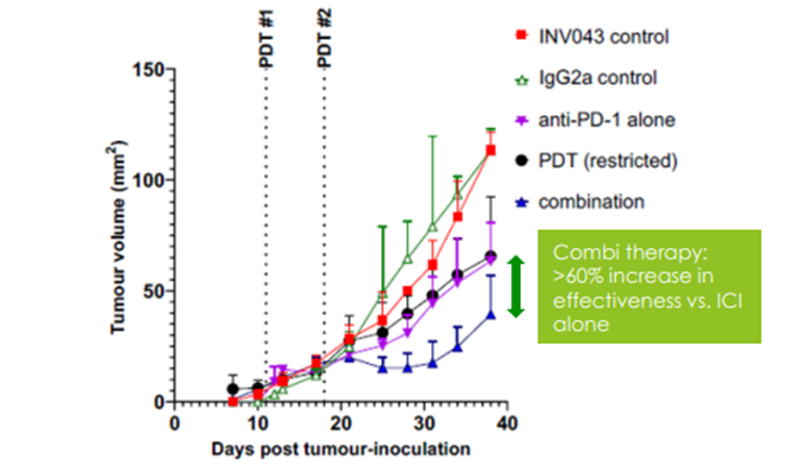
Image source: IVX update
Image description: Results of INV043 in combination with ICI
Having achieved strong in vivo and in vitro results across several types of cancer, Invion plans to conduct clinical trials as the next step. The indications for these trials would cover skin cancer, anogenital cancer, and other solid tumour cancers.
Major developments in infectious diseases research
Invion is exploring the potential applications of PhotosoftTM in infectious diseases. The company witnessed positive screening results from in vitro study of PhotosoftTM technology against the Zika virus. PhotosoftTM compounds showed more than 99% inhibition against Zika virus in cell-based assays following the application of light.
PhotosoftTM also displayed positive screening results from in vitro studies on the inhibition of the Dengue virus. Eight out of ten compounds in the test showed antiviral activity against the Dengue virus on exposure to light. Those eight compounds also had superior Therapeutic Index (TI) values compared to Monensin (an antibiotic against the Dengue virus).
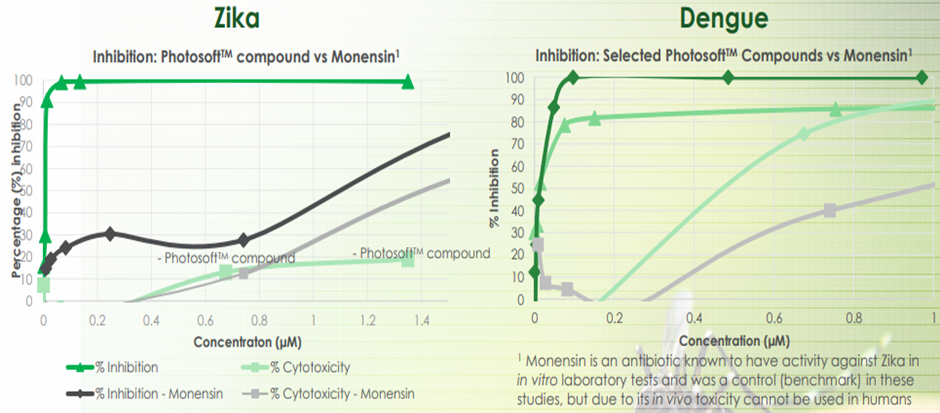
Image source: IVX update
In another test against infectious diseases, PhotosoftTM compounds showed broad-spectrum anti-microbial potential. In vitro tests conducted by leading institutions marked the effective potential of PhotosoftTM against several types of pathogens (viruses, bacteria, and fungi), including antibiotic-resistant superbugs.
Further, PhotosoftTM compounds were also found to be effective against SARS-CoV-2, the virus that causes COVID-19. Multiple PhotosoftTM compounds showed effectiveness against the Delta and Omicron variants of COVID-19 at far lower concentrations than Remdesivir during in vitro tests. The compounds exhibited either no or low cytotoxicity.
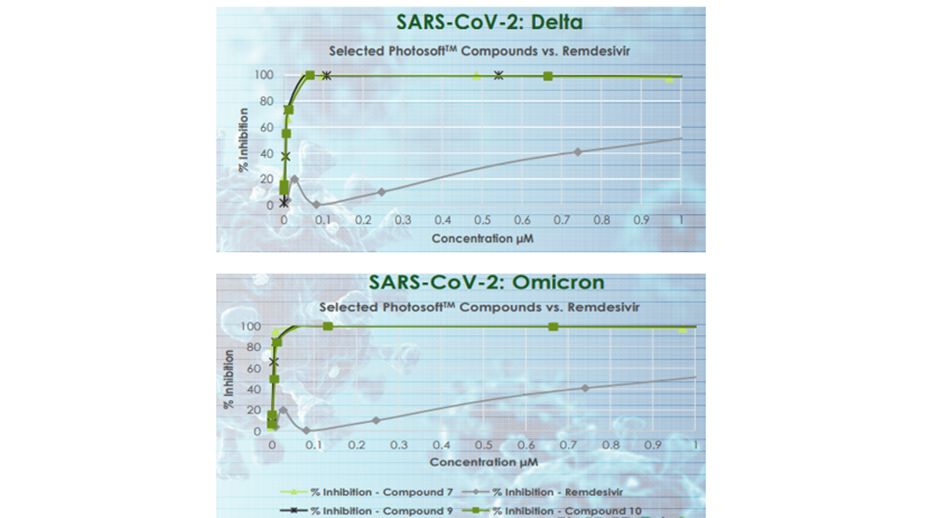
Image source: IVX update
Moving forward in infectious diseases, Invion plans to identify target indications and carry out PoC on target indications.
Vast market opportunities
Invion says that its research in multiple indications provides exposure to large addressable markets worth billions of dollars. Moreover, they open avenues for partnership opportunities.
Below is a quick snapshot of global market opportunities for some of the indications:

© 2022 Kalkine Media®, Data source: IVX update
IVX shares closed at AU$0.011 on 18 November 2022, up nearly 5% from the last close.







