Botanix Pharmaceuticals Ltd (ASX: BOT) is a dermatology company. It is based in Perth, Australia and Philadelphia, PA. The company aims to develop safe and effective topical treatments for skin conditions like acne, psoriasis and atopic dermatitis. It also treats inflammation, deterioration of the skin barrier, skin cell proliferation, pruritus, excess sebum production and bacterial infection. The company uses a natural compound known as cannabidiol or CBD in its products.
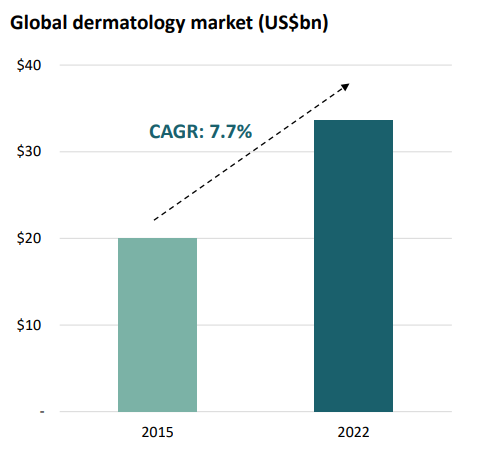
The Global dermatology market (Source: Companyâs report)
FACT: According to the Koncept Market Research, the dermatology global market is expected to expand to US$33.7bn by 2022, and the pipeline of new products remains impossibly thin.
BOT undergoes fast clinical development strategy to pace its product commercialisation. Its clinical studies are usually completed in a time frame of 4 to 12-weeks.
The company has a license to use PermetrexTM for direct skin delivery of active production of medicines in skin diseases. The company is working with various parties to test its applications, on fee-for-service and traditional license basis.
In early 2018, BOT completed its first acne patient studies with BTX 1503. Phase 2 of the study is expected to be released in Q3 2019. Another, Phase 2 study in atopic dermatitis is in process and is likely to be done before the end of 2019. The Phase 1b BTX 1308 psoriasis mechanism of action of CBD study, would be available in 2Q CY2019. Other pipeline projects of BOT, related to cannabidiol, are moving ahead and are planned to be in the clinic in 2H 2019.
On 20th May 2019, the company announced that it had appointed Mr Vince Ippolito as the companyâs new President. He is a US-based dermatology industry leader and bears an experience of over 30 years in the pharmaceuticals industry, and almost 20 years exclusively in dermatology. Besides, he is well known for his contribution towards the recent sale of dermatology company Anacor to Pfizer for US$5.2bn.
Prior to joining BOT, Mr Ippolito was President and Chief Operating Officer of Dermavant Sciences. Dermavant is engaged with the treatment of both- inflammatory skin diseases condition and conditions related to medical dermatologic. It recently completed a US$330m transaction with GSK. Apart from this, Mr Ippolito is also part of the Board of Suneva Medical, which is a private regenerative aesthetics company.
He has also been associated with Anacor Pharmaceuticals, where he built the marketing and sales functions and developed the companyâs product portfolio. In the early part of his career, he has launched multiple new dermatology products at Medicis Pharmaceutical Corporation. He later served as Senior Vice President, General Manager, Aesthetics.
As BOTâs President, Mr Ippolito would be taking care of the companyâs commercial operations worldwide. He would also join the companyâs Board as Executive Chairman.
Also, BOT had issued 3,000,000 unlisted options on 20 May 2019, exercisable at $0.115 on or before 20 May 2023, under its Employee Securities Incentive Plan to him, which would expire in 4 years from their date of issue. The Unlisted Options are being issued under the Companyâs Employee Securities Incentive Plan to provide an incentive for future performance.
Share Price Information:
The market capitalisation of the company is A$87.99 million. The 52-week high and low of the stock is A$0.190 and A$0.067, respectively. The stock of the company closed flat at A$0.115 (as on 22 May 2019). In the last one year, the stock has delivered a return of -14.81%, and the YTD return stands much better at 57.53%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.







