Summary
- COVID-19 pandemic is being considered as the Great Virus Crisis (GVC), having taken many lives and no proven vaccine or treatment yet.
- Several companies across the globe are in a race to develop a treatment or a vaccine for the disease. Australian biotech player CSL Limited is one of them.
- CSL Behring has joined the COVID-19 fight and to provide Immunoglobulin for treatment.
- The Company also entered a collaboration Thermo Fisher Scientific to offer best-in-class pharma services and partnered with BrightInsight to improve treatment options for rare diseases.
The healthcare sector, over the last 12 months, has been one of the best performers on the Australian Securities Exchange (ASX). Some players from the healthcare industry in Australia have witnessed a substantial increase in share price in the current times, despite the market fluctuations that have taken place during the turmoil.
The COVID-19 crisis has generated opportunities for the healthcare sector. The biotech players have accelerated their R&D efforts to develop a vaccine for this respiratory disease. Further, telemedicine has garnered a lot of attention has this indicates that the companies need to move towards digital healthcare and focus on virtual care services.
In this article, we will highlight one ASX-listed leading biotech player- CSL Limited. Let us see what’s the latest development for this biotech behemoth.
About CSL Limited
ASX-listed Melbourne-headquarters leading biotech company (ASX:CSL) is engaged in the development and delivery of innovative biotherapies and influenza vaccines to save lives and provide support to patients having life-threatening medical conditions. The Company has its operations in more than sixty countries with more than 26,000 employees across the globe. With eight manufacturing sites across six countries Australia, China, the UK, Germany, Switzerland, and the US. CSL has over 257 plasma collection centres across North America and Europe.
How is CSL Responding to COVID-19?
CSL Limited, the largest biotech company in Australia, stated that it is working on a plasma-based treatment for COVID-19 patients having a severe infection. The Company highlighted that it is providing its technologies, expertise, equipment along with materials to offer support in response to COVID-19.
Notably, CSL is providing additional southern hemisphere influenza vaccine doses across Australia during the COVID-19 crisis, and the Company has implemented business continuity plans across its all sites.
The below-mentioned image represents how the Company is contributing to combating the pandemic:
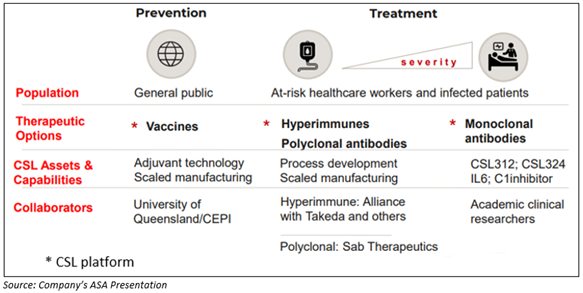
INTERESTING READ: Healthcare Company CSL beats CBA and BHP as the most valuable company
CSL Behring Begins Development of Treatment for Severe COVID-19
CSL Behring disclosed that it is engaged in developing the plasma-derived therapeutic that has potential to treat severe complications of COVID-19.
On 6 May 2020, the biotech division of CSL Limited revealed that it would commence immediate onshore manufacturing of an anti-SARS-CoV-2 plasma product which has the potential to treat patients with severe COVID-19 complications, especially those who are requiring ventilators for treatment.
CSL notified that the investigational drug would be identified as COVID-19 Immunoglobulin, and this will be manufactured by CSL Behring Australia at its manufacturing facility in Broadmeadows, Victoria.
The Immunoglobulin would be manufactured by using plasma donated by Australians who get recovered from the coronavirus infection. As per the estimation near about 800 plasma donations will be needed to develop adequate Immunoglobulin for providing treatment to 50-100 seriously ill patients under the clinical study.
COVID-19 Immunoglobulin development from plasma donated in Australia is one of several methods CSL is exploring to provide support in fighting against the COVID-19 pandemic.
Moreover, the Company is part of a formerly named world-leading global alliance formed with other companies that are engaged in the manufacturing of plasma products.

After the successful completion of the clinical study and subject to a request by the governments of Australia, CSL Behring would apply for registration of COVID-19 Immunoglobulin with the Therapeutic Goods Administration (TGA) to get permission for its ongoing supply in Australia.
CSL would work with the National Blood Authority (NBA) for development of Immunoglobulin to combat COVID-19 for people across Australia.
CSL Boosts Its Business by Collaborating with Thermo Fisher Scientific
On 27 May 2020, CSL revealed that the Company had entered a strategic partnership with the world leader in serving science, Thermo Fisher Scientific. This partnership is for the lease of CSL’s state-of-the-art biotech manufacturing facility that is currently under construction in Switzerland along with the contracting of capabilities and capacities from within the Thermo Fisher network.
This follows a strategic review by CSL to establish a pathway that would completely enhance the capabilities of the Lengnau facility after completing the construction in 2021.
The agreement between both the companies is long term and includes Thermo Fisher leasing and operating the Lengnau facility of CSL and being responsible for providing production to support biologics portfolio of CSL, along with a variety of other contract manufacturing services like packaging, fill-and-finish for several products of CSL.
CSL’s New Debt Facilities to Strengthen Maturity Profile
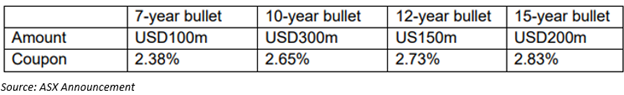
On 7 May 2020, CSL revealed it had priced a new US$750 million private placement a day earlier. Notably, this new debt facilities would strengthen the current debt maturity profile of the Company and was well-oversubscribed by its investors. The private placement by CSL comprises four maturities:
CSL Behring Partners with BrightInsight to Improve Treatment for Rare Diseases
On 6 May 2020, CSL Behring collaborated with BrightInsight which provides a leading global regulated IoT SaaS platform for MedTech and biopharma. With this partnership, CSL Behring is leveraging platform regulated by BrightInsight for managing and providing support to its digital health portfolio focused on providing treatment for rare and severe diseases.
CSL Behring will build as well as host digital health tools, algorithms, applications, and medical software (SaMD) on the platform of BrightInsight. By using this platform, CSL could focus on therapeutic innovation instead of the underlying technology.
Stock Performance: on 29 May 2020, CSL stock closed market at AU$276.220, down by 3.927% compared to the previous close. The Company has a market cap of AU$130.54 billion, with ~454.05 million outstanding shares.



