Source: PhotobyTawat, Shutterstock
Summary
- Shares of AstraZeneca have experienced a turbulent trading session of late.
- The stock was trading at GBX 7,133, up 0.07 per cent on 19 March.
- This year so far, the stock has largely fallen extending the previous year’s trend.
Shares of Cambridge-headquartered Pharmaceutical giant AstraZeneca Plc (LON: AZN) have experienced a turbulent trading session of late after the news reports surfaced that indicated adverse effects of the Covid-19 vaccine jointly developed by the company and the University of Oxford.
Inverse head and shoulders pattern
An inverse head and shoulders pattern in making has been witnessed in the AstraZeneca shares, looking at the trading activity in the last couple of weeks as the share price reacted evidently to the vaccine chaos with more than a dozen of nations discontinuing the inoculation programme through AstraZeneca jab.
According to the data available with the London Stock Exchange, the shares of AstraZeneca rose for five consecutive sessions, rising as much as 5.86 per cent between 3 and 10 March, making a high of GBX 7,192 per share.
AstraZeneca shares (5 March - 18 March)
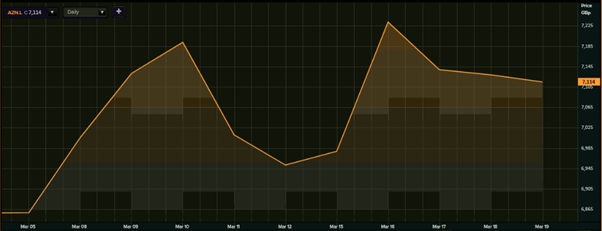
(Source: EODHD/Others, Thomson Reuters)
In the next two sessions after 10 March, the stock slipped 3.35 per cent, touching a bottom of GBX 6,951 on 12 March. The stock then bounced back to GBX 7,232 after two sessions on 16 March registering a gain of a little more than 4 per cent. Barring present day’s share price action, the stock has fallen 1.44 per cent to GBX 7,128 since then.
AstraZeneca shares traded little changed on Friday, 19 March, oscillating between a high and low of GBX 7,155 and GBX 7,092, respectively. At around 1146 GMT, the stock was trading at GBX 7,133, up 0.07 per cent from the previous close of GBX 7,128.
AstraZeneca shares (YTD)
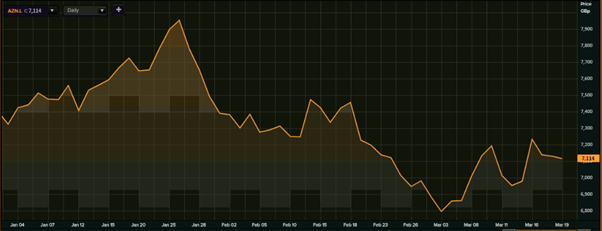
(Source: EODHD/Others, Thomson Reuters)
So far in this calendar year, the stock of AstraZeneca has largely fallen, extending the previous year’s trend after the end of July 2020. The stock is down 2.68 per cent on a year-to-date (YTD) scale, while it has plunged more than 10 per cent from the YTD high of GBX 7,952 (26 January 2021).
The backdrop
In certain parts of Europe and Africa, there were cases of apparent blood clotting after the administration of the Oxford-Astrazeneca Covid-19 vaccine. Following the development, a couple of countries have halted the distribution of Astrazeneca Covid-19 jab.
The evidently faulty roll-out of the vaccine in the ongoing immunisation drive across the world has once again renewed the tensions amid the respective healthcare administrations in many nations.
Nonetheless, the UK Medicines and Healthcare products Regulatory Agency (MHRA) reaffirmed that people should continue to receive the vaccine doses as the prospective benefits far outweigh the adverse reactions or the apparent risks associated with the vaccine.
The blood clots in the veins are not caused by the AstraZeneca-Oxford Covid-19 vaccine, MHRA concluded after carrying out a scientific review of all the available data on 18 March.
The Commission on Human Medicines, the independent advisory group to the UK government, has also confirmed the same after its clinicians reviewed the data. The MHRA has separately reviewed the data of Covid-19 cases who have been given the AstraZeneca jab from the hospital admissions and the records held with respective GPs.
Furthermore, the MHRA is supervising a detailed review with regard to a specific type of blood clot in cerebral veins as reported in five cases in the UK. According to the regulator, the very rare type of blood clot has been reported in less than 1 person out of a million individuals who have received the AstraZeneca jab in the UK. The association of blood clotting with the vaccine administration has not been established so far.
More than a dozen of countries that temporarily banned the distribution of AstraZeneca Covid vaccine include France, Germany, Italy, Luxembourg, Netherlands, Norway, Sweden, Austria, Bulgaria, Denmark, Estonia, Iceland, Ireland, Latvia, Lithuania, and Romania from Europe. Congo, Indonesia, South Africa, Thailand, and Venezuela were the countries outside Europe that suspended the distribution.
.jpg)


_06_04_2026_00_29_48_108019.jpg)


