A biotechnology company listed on the Australian Stock Exchange (ASX), Imugene Limited (ASX: IMU) develops cancer immunotherapies targeting B-cell peptide vaccines. The immunotherapies developed by the company seek to activate the cancer patientsâ immune system in order to treat and eradicate tumors. Imugene is a leader in the B cell peptide cancer vaccine space and has a developing pipeline of cancer vaccines, including B-Vaxx (anti-HER-2), HER-Vaxx (anti-HER-2) and PD1-Vaxx (anti-PD-1). Besides this, the company is testing a combination of HER-Vaxx and PD1-Vaxx that is in an early stage of development.
New Positive Clinical Data Presented at ESMO
In an announcement on the ASX today, Imugene presented new positive clinical data of IMU-131 HER2/neu peptide vaccine plus chemotherapy, from the Phase Ib study of its HER-Vaxx cancer vaccine ,at the European Society of Medical Oncology (ESMO) World Congress on Gastrointestinal Cancer, held in Barcelona, Spain over the weekend.
IMU-131 HER2/NEU peptide vaccine is a B-cell peptide vaccine that is given to patients with HER2/neu overexpressing Gastroesophageal Junction (GEJ). The vaccine attacks the same target as the cancer drug Herceptin®.
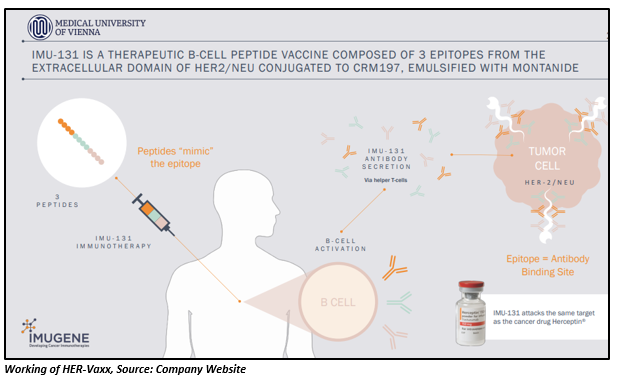
The new clinical data from the Phase Ib study of Imugeneâs HER-Vaxx cancer vaccine shows that the patients receiving the optimal biological dose of vaccine continued to pose positive cancer-fighting antibody and clinical response rates. The data was presented by Dr Marina Maglakelidze, a professor from the Research Institute of Clinical Medicine located in Tbilisi, Georgia, through an oral presentation.
According to Managing Director and Chief Executive Officer of Imugene, Leslie Chong, this latest presentation further strengthens the excellent early results and ongoing interest in companyâs potentially promising B-cell platform cancer vaccine among the international oncology community.
Clinical Data Results
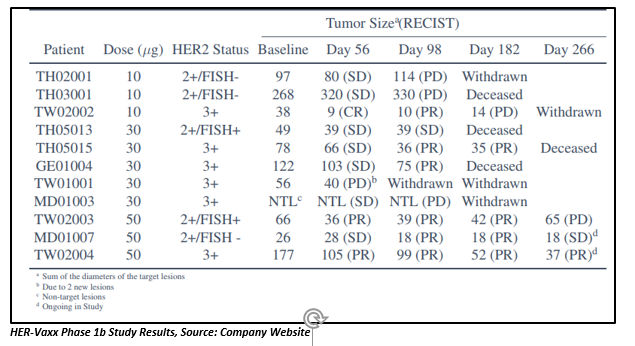
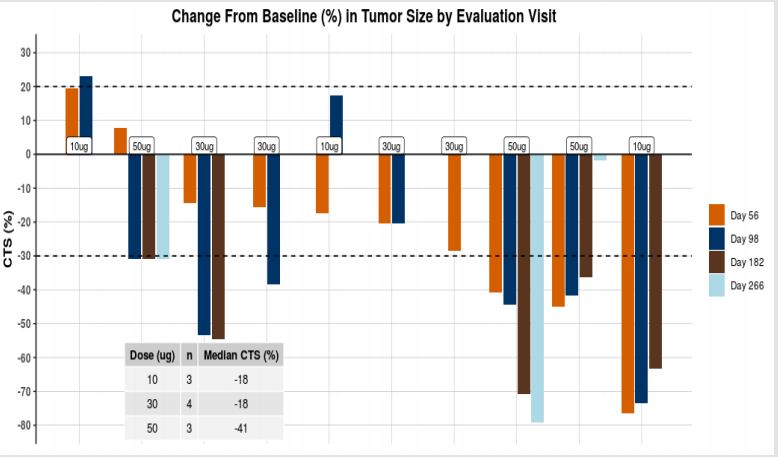
Strong and high levels of HER-2 targeting antibodies have been maintained by the patients, signifying a presence of durable response with no resistance developed. Among cohort 3 patients, one patient has observed a total tumor reduction of almost 80 per cent from baseline CT scan at Day 266 visit, with 1 out of 4 of their lesions becoming unmeasurable by RECIST criteria.
Phase Ib study Safety Results
No DLTs were observed in the study, and there were no such events that led to study drug discontinuation. In all, 207 TEAEs (Treatment emergent adverse events) were reported by fourteen patients with most of the events assessed as Grade 1 to 3 severity and were not associated with IMU-131. The TEAEs observed were expected for the chemotherapy the patients received.
One patient in the IMU-131 50 µg treatment group reported two Grade 1 vaccination site reactions (pruritus and erythema), which were assessed as possibly related to IMU-131. Two TEAEs - hypoalbuminemia and hyponatremia were reported by one patient in the 30 µg group that were assessed as related to IMU-131. Another patient in the 30 µg group reported two TEAEs (decreased appetite and weight decreased) that was also assessed as possibly related to IMU-131.
Source: Company Website
Phase Ib Study Design
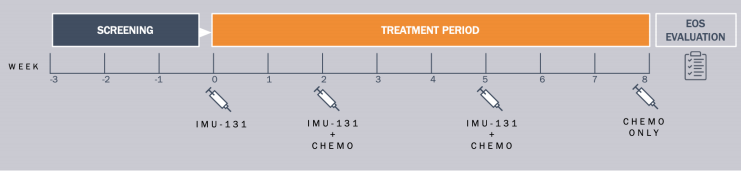
14 patients were enrolled for the Phase Ib study from 14 sites in 5 countries that were divided into 3 cohorts with three patients in 10 µg cohort, six in 30 µg cohort and five in 50 µg cohort. The doses are being given in combination with chemotherapy. The enrolment of the metastatic gastric cancer patients for the Phase 1b HER-Vaxx study was completed in late 2018.
Under the study, gastric cancer patients (stage III or IV gastric or GEJ adenocarcinoma) with 10 HER2 overexpressing tumors (3 x HER2++ FISH positive, 7 x HER2+++) and 4 HER2++ expressing tumors are being given a dose level of 10 µg, 30 µg and 50 µg of IMU-131 on days 0, 14, and 35; accompanied by 5-fluorouracil or capecitabine and cisplatin every 21 days.
Source: Company Website
Previous Results
No safety or toxicity issues were reported; all patients reported increased antibody responses. The Phase1b trial results showed that out of the 14 patients enrolled in the study, eleven were evaluable for tumor progression at day 56 and later. Of those patients, one patient saw the complete response, four recorded a stabilisation of their disease, five had a partial response and one had a progressive disease. There were no SAEs related to HER-Vaxx, and one patient had a mild injection site reaction.
Recently on 4th June 2019, the company also presented positive data of its HER-Vaxx vaccine at the Annual meeting of prestigious ASCO (American Society of Clinical Oncology) held in Chicago Illinois, the United States from 31st May to 3rd June 2019. Imugene informed that the tumors of patients who received the highest dose of HER-Vaxx have either shrunk further or stabilized by day 182 of the trial. It was observed that the tumor shrunk more than 70 per cent in one patient relative to the tumor size at the time of joining the trial.
What do HER-Vaxx Previous Results Indicate?
The preliminary immunology and clinical response data of HER-Vaxx are promising. The pre-clinical study and the Phase I study results of HER-Vaxx shows that the vaccine stimulated a potent polyclonal antibody response to a well-known and validated cancer target, HER-2/neu. The safety data demonstrated that the vaccine is well-tolerated with no significant local or systemic reactions. No dose-limiting toxicities, significant injection site reactions and IMU-131 related SAEs were observed in the patients. Also, the preliminary response data indicated that 50 µg of IMU-131 was associated with tumor size reduction. The 50 µg of IMU-131 produced the most consistent anti-peptide (P467) and anti-HER-2 specific antibodies compared to 10 and 30 µg doses.
Imugene advanced to Phase 2 study when the objectives of Phase 1b study were met as all the dose levels indicated an increased antibody levels in all patients. The first patient was dosed with IMU-131 cancer vaccine in Phase 2 study in March this year. Imugene is using 50 µg dose of IMU-131 in the Phase 2 study.
Stock Performance
Imugeneâs stock is currently trading higher on ASX at AUD 0.016 (on 8th July 2019), up by 6.67 per cent relative to the last closed price. Around 5.3 million shares were traded in the session today as compared to its annual average volume of 3.7 million.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. The above article is sponsored but NOT a solicitation or recommendation to buy, sell or hold the stock of the company (or companies) under discussion. We are neither licensed nor qualified to provide investment advice through this platform.







