Highlights
- Bile tract cancer expansion study opens after the completion of the high dose cohort in the MAST trial.
- This new trial aims to enrol 10 patients with bile tract cancers, specifically cholangiocarcinoma.
- Interim findings from the MAST trial have revealed encouraging responses in gastrointestinal cancers, especially in cholangiocarcinoma.
- The clearance of the fifth cohort in the intratumoural (IT) arm of the VAXINIA monotherapy trial marks a significant milestone.
Imugene Limited (ASX: IMU) has commenced enrolment for the expansion study focusing on cholangiocarcinoma patients. This comes after the completion of the fifth cohort in the intratumoural (IT) arm of the monotherapy dose escalation trial assessing the company’s CF33-hNIS (VAXINIA), a cancer-fighting virus.
The MAST (Metastatic Advanced Solid Tumours) Phase 1 trial will be expanded to enrol 10 individuals diagnosed with bile tract cancers. This follows encouraging initial responses observed in gastrointestinal cancers, especially in cholangiocarcinoma.
Cholangiocarcinoma is a rare condition in which malignant cancer cells are created in the bile ducts. It has been characterised as difficult to treat disease and shows limited response to immunotherapy medications.

Initial encouraging responses observed in cholangiocarcinoma
A cholangiocarcinoma patient who had failed three previous lines of therapy was administered a mid-dose of VAXINIA as monotherapy via IT injection. Remarkably, this patient achieved a complete response, indicating the disappearance of any detectable signs of cancer following treatment, and has showed no recurrence for over 430 days.
Another patient with cholangiocarcinoma who had also undergone prior drug therapies showed stable disease for over four months after IV-administration of VAXINIA.
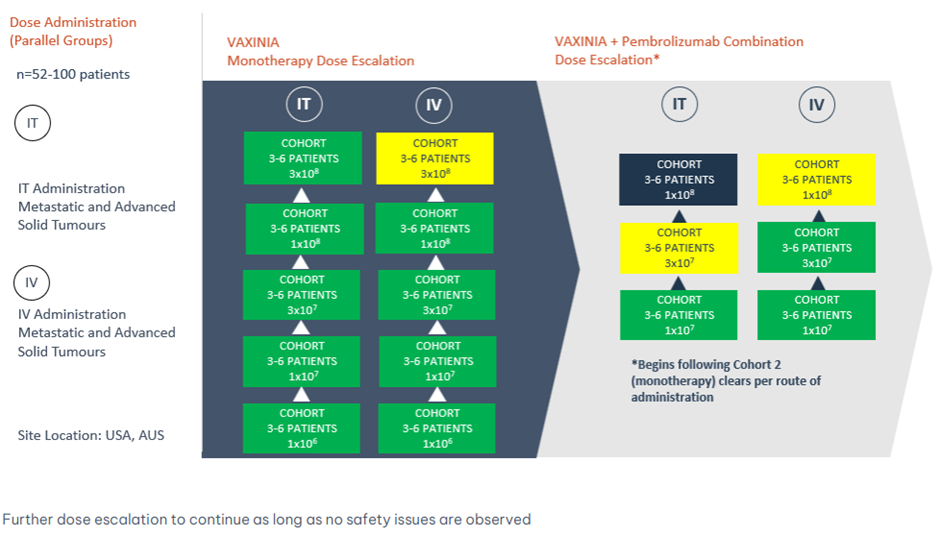
image source: company update
Background of the trial
The trial was granted the Fast Track Designation by the FDA for bile duct cancer treatment in November last year. This designation enables Imugene to collaborate more closely with the FDA, expediting the program and potential approval process. It comes in response to the promising Phase 1 data showcasing efficacy and tolerability.
on 12 April 2024, the Cohort Review Committee gave the green signal to the fifth cohort in the IT arm of the monotherapy dose escalation portion of the MAST trial, with no safety signals detected till date.
The company is also advancing enrolment for the pembrolizumab and VAXINIA combination of the study with 16 patients dosed to date.
IMU shares traded at AU$0.092 apiece at the time of writing on 15 April 2024.



