Highlights
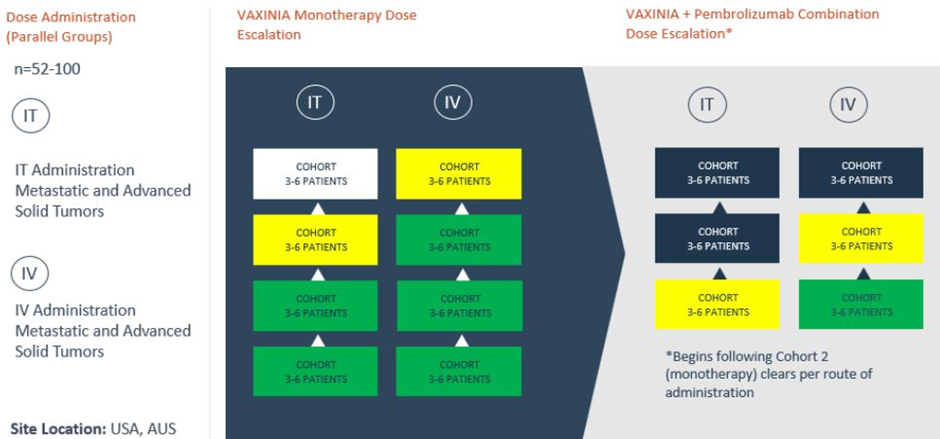
- Phase 1 MAST trial of VAXINIA is related to patients suffering from metastatic or advanced solid tumours
- The trial has cleared cohort 3 and entered cohort 4 of intravenous (IV) monotherapy dose escalation
- The trial has cleared cohort 1 and entered cohort 2 of intravenous combination with pembrolizumab therapy dose escalation
- VAXINIA is stated to be a novel cancer-killing virus, which has been developed by City of Hope
Listed immuno-oncology company Imugene Limited (ASX: IMU) has informed that the VAXINIA MAST trial (Phase 1) has progressed to the next cohort in both the intravenous monotherapy and combination studies. It is pertinent to note that the trial relates to metastatic advanced solid tumours, and it evaluates the safety of VAXINIA, a City of Hope-developed novel cancer killing oncolytic virus.
Imugene notes that the multicentre Phase 1 trial has cleared cohort 3 of the monotherapy arm and cohort 1 of the combination arm. The company's MD & CEO has lauded the progress and commented that IMU is aiming for peer review and recognition of the science in publications and conferences.

Source: Company update
Shares jump
The company’s ASX-listed shares were trading in the green zone at a price of AU$0.100, up over 7.5%, after the cohort advancement announcement.
More
Imugene Limited's VAXINIA MAST (metastatic advanced solid tumours) trial has advanced into the next cohort.
Cohort 3 in the Phase 1 trial has been cleared, with cohort 4 (intravenous arm of the monotherapy dose escalation) now opened.
Besides, the company has informed that cohort 1 of the intravenous arm (combination study with Pembrolizumab) is cleared. This allows the study to progress to cohort 2.
Source: Company update
Notably, VAXINIA’s Phase 1 is a multicentre trial. It involves administering a dose of City of Hope-developed cancer-killing virus CF33-hNIS to patients who have had a minimum two previous lines of standard of care treatment. It is stated by IMU that the virus has exhibited the potential to shrink various types of cancer tumours during preclinical lab and animal models.
The ongoing clinical trial of VAXINIA began in May last year. It will likely run for nearly 24 months, with funds being deployed from existing budgets and resources, IMU notes. The study has the objective of recruiting up to 100 patients in the multicentre (~10 sites) trial to be conducted in Australia and the US.
About Imugene
The immuno-oncology company -- developing immunotherapies that can demonstrate the ability to activate cancer patients' immune system for the treatment of tumours -- is listed in Australia on the ASX. Imugene states it is focused on harnessing the affected person’s immune system against tumours.
IMU’s pipeline include numerous immunotherapy B-cell vaccine candidates. It also has an oncolytic virotherapy (CF33) aimed at treating a variety of cancers in combination with standard of care drugs and emerging immunotherapies such as CAR T’s for solid tumours.



