With the recent outbreak of the novel coronavirus (2019-nCoV) in Greater China, the healthcare space has gained massive traction alike from public authorities and private investors. Healthcare specialists have been exposed to a challenge to invent a robust cure to the virus, that has claimed over 1000 lives and confirmed cases have increased to nearly 43,000, globally.
ASX-listed stocks have been responding to the situation in several ways. Let’s take a couple of recent examples-
- Global leader in implantable hearing solutions, Cochlear Limited (ASX:COH) has recently reduced the guidance outlook for FY20 from $290-300 million to $270-290 million, with hospitals across Greater China currently deferring surgeries, including cochlear implants, to limit the risk of infection from the coronavirus.
- On a different note, focused on research, development, and commercialisation of drugs targeting significant viral diseases with unmet medical need, Biotron Limited (ASX:BIT) recently announced that it is in the process of evaluating several promising compounds for activity against coronavirus.
Besides the Coronavirus debacle, ASX-listed healthcare companies have been making headlines for tapping milestones and with business updates. We have cherry-picked two stocks with recent updates-
Starpharma Achieves Milestone Under Multi-Product License with AstraZeneca
Starpharma Holdings Limited (ASX:SPL) is a world leader in the development of dendrimer products for life science, pharmaceutical and other applications. The Company’s underlying technology is built around dendrimers. Besides this, it has two core development programs: VivaGel® portfolio and DEP® drug delivery. SPL also develops several products internally and others via commercial partnerships.
On 11 February 2020, the Company notified that it had received $4.5 million from AstraZeneca under its multi-product license. As per the license, SPL was eligible to receive development, launch and sales milestones (till USD 124 million) along with tiered royalties on net sales for the product (here, AZD0466) and USD 93.3 million for each subsequent qualifying product.
The milestone has been received post the successful dosing of the first patient in the phase 1 clinical trial of AZD0466, which is believed to be a ‘best-in-class’ agent with a broad opportunity in solid and haematological tumours, given its ability to target both Bcl2 and Bcl/xL. The trial is being conducted at various sites across the US and will recruit patients with an array of cancers. It was the first patient dosed in the phase 1 trial that had triggered the USD 3 million milestone.
Alcidion Group’s Business Update
A tech-savvy healthcare company with over 25 years of combined healthcare experience, Alcidion Limited (ASX:ALC) aims to transform the healthcare space with its smart, intuitive technology solutions. The Company offers a complementary set of software products and services that enable task and communication management, boost interoperability, and provide clinical decision support to the healthcare space.
ALC believes that of late, healthcare providers have been facing a few challenges pertaining to industry (disenfranchised workforce and budget pressure), demographic trends (ageing population and rise in chronic disease), rising costs and risks (increasing penalties for unsafe care and poor outcomes).
In the need of the hour, they should adopt technology to improve patient outcomes and deliver efficiencies to meet demand. The Company also believes that the global market for clinical decision support systems (CDSS), specifically, is likely to grow rapidly, 39 per cent over five years to USD 2.8 billion.
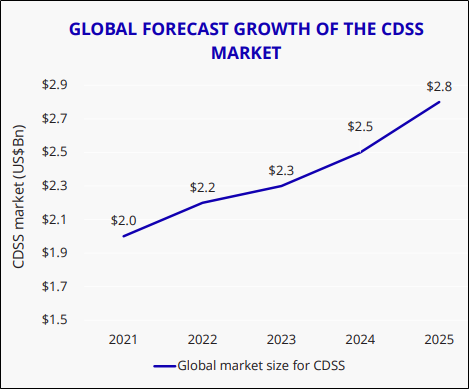
(Source: ALC’s Presentation)
On 11 February 2020, Alcidion Group released an investor/ business update, to be presented to investors this week by Managing Director Kate Quirke. Below are a few highlights from the same-
Miya MEMRe- Mobile EMR + CDSS
- ALC offers a unique technology platform Miya MEMRe, which is a real-time, CDSS platform providing smart infrastructure for healthcare. The next generation CDSS will be able to support a high volume of algorithms and respond to changes in patient status, while the automation improves productivity and ensures better accessibility of critical information
- The solution is the first locally developed fully mobile EMR solution, offering the full benefits of Miya Precision on mobile devices
- It uses Miya Precision Natural Language Processing (NLP) and is based on the open FIHR standard
Market Opportunity
The United Kingdom
The Company acknowledges the fact that there has been heavy investment in digital health across all markets. It also believes that UK, an early adopter of new technology, is a large market opportunity. The region is likely to have a favourable operating environment post-election, with investments promised by policymakers to support the entire NHS achieve digital maturity.
ALC believes that the existing Patientrack base in the UK provides a solid footing for cross-sell. Over the next 5 years, the total market potential in UK for all products and services is USD 1.1 billion and ALC’s target accessible market in UK, adjusted for achievability is estimated at USD 586 million.
Tapping the UK opportunity, ALC aims to-
- Scale up marketing function and sales force, with new GM appointed in Q2 and a number of planned sales staff appointments
- Advance integrated EMR and add value to existing EMR implementations
- Focus on penetration and growth via replacement of paper and pagers as per NHSX strategy
- Offer a smart healthcare platform for interoperability
- Commercially launch Miya MEMRe to UK market in Q3 FY20
Australia and New Zealand
Closer to home, ALC has a robust Australian growth strategy in place. It aims to add value to existing EMR investment via mobile EMR and clinical decision support and Offer alternative to EMR. The Company also aims to Use results from recently completed Proof of Concept (PoC) for Miya MEMRe to drive adoption of the platform in pilot sites.
Besides this, ALC will continue to scale up marketing and sales function across the ANZ region, with new staff appointments and use data analytics services to build clinical data warehouses as platform for decision support software. The Company will also target integration services at strategically important initiatives.
According to ALC, the total market potential in NZ for all products and services is $75 million and ALC’s target accessible market in NZ, adjusted for achievability is $42 million.
Corporate Highlights
Through a successful institutional placement in November 2019, ALC raised $16.2 million to accelerate growth and strategic expansion. It has made a solid start to FY20 with strong sales and several investments made to scale up the business. Q2 FY20 highlights include-
- $15.4 million revenue currently able to be recognised in FY2020
- Total sold revenue of $37.2 million out to FY2025
- Strengthened cash reserves of $17.2 million
- New contracts sold in Q2 totaled $3.5 million
- Two contracts signed in January 2020 with Calvary Health Care and eHealth NSW, adding a further $1 million to the sold revenue of Q2
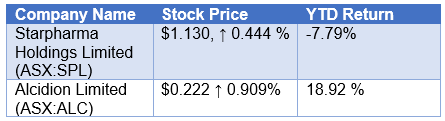
Stock Performance
Let us look at the stock performance of the discussed stocks, during the market session, as on 12 February 2020 (at AEDT 2:23 PM)-