In Australia, there are plenty of high-quality health care companies, most of which have delivered excellent returns. Investment in health care stocks is high yielding; however, it does include risks. If undergoing clinical studies fail to deliver the desired results or if there are obstacles to securing regulatory approvals, the share price might report a drop. Stocks in the health care sector that are approaching towards the completion of their research & development stage or awaiting regulatory approval are also expected to deliver better returns to the investors.
Here we are discussing two ASX listed health care stocks, which have planned product development and R&D.Biotron Limited (ASX:BIT)
Health care sector player based in Sydney, Australia, Biotron Limited (ASX:BIT) is a developer of drugs, designed to treat viral diseases with an unmet medical need. The company is also into commercialisation activities.
The company is focused on novel small molecule approach targeting viroporin proteins, which are important to facilitate the pathogenicity of hepatitis C, HIV-1, Dengue, Zika, Influenza and other viruses. Currently in the second phase development, BIT225 is the lead compound of the company, being developed to treat HIV-1 & hepatitis C. The viroporin approach has shown early in-vitro activity against Dengue, Zika and other viruses.
September Quarter Update
On 21 October 2019, the company released its quarterly report for the period ended 30 September 2019, unveiling net cash outflow towards operating activities of 881,000 and net cash inflow from financing activities of $5,000. Cash and cash equivalents at end of quarter stood at $4.87 million. For the next quarter, the company is expecting cash outflows of $1.84 million.
Expert Scientific Advisory Board
- On 1 October 2019, the company announced about establishing an expert Scientific Advisory Board (âSABâ), which will be involved in the core Phase 2 HIV-1 clinical development program.
- Expert Scientific Advisory Board is an international group of experts with interdisciplinary experience within the HIV-1 field, covering clinical developmental aspects of HIV-1 therapies.
- Chairperson for SAB will be Professor Robert L. Murphy, who is Executive Director, Institute for Global Health at Northwestern University Feinberg School of Medicine.
Key Highlights of FY2019 ended 30 June 2019
- Biotron achieved significant milestones during the year from successful outcomes of the BIT225?009 phase 2 HIV?1 clinical trial, which were released to the market in late 2018.
- The company presented data from the trial at the HIV DART and Emerging Viruses 2018 Conference in the US.
- BIT appointed Professor Stephen Locarnini as a Non-Executive Director and raised $6.0 million in capital.
Hepatitis B Virus Program Update
Treatment and control of hepatitis B virus has been a crucial focus of endeavour for antiviral drug development by health care industries. The number of people infected with HBV has grown to 2 billion, globally. According to WHO estimates, over 250 million people are chronically infected.
Biotron came up with an approach of novel small drug molecules which effectively treat HBV in cell cultures. The company is working with a US-based research organisation for characterising the anti-HBV activity of its compounds.
Clinical Development
BIT225, the lead compound of Biotron, is in the mid-stage of its clinical development, targeted towards HIV-1 and Hepatitis C virus (HCV) infection. The company has completed seven clinical trials with the compound. A total of 55 healthy volunteers and 94 infected subjects either with HIV-1, HCV or co-infected with HIV-1/HCV, have received the BIT225 treatment.
Clinical Trial Results for BIT225
For HIV-1
- The aim of BIT225, a novel first-in-class HIV-1 Vpu protein inhibitor; is inhibition of virus replication in cellular reservoirs.
- In vitro studies on BIT225 have shown that it significantly inhibits HIV-1 release in a dose-dependent manner from CD14+ and CD16+ monocyte subsets at various stages of differentiation and monocyte-derived dendritic cells (MDDC)
- The outcomes of studies in the clinic-
- Reduction of HIV-1 release from CD14+ monocytes observed from BIT225 treated isolated cells from HIV-1 infected patients
- The compound may impact on immune activation, which is associated with macrophage.
- BIT225 is permeable to Blood-brain barriers (BBB) and has the potential to treat HIV-associated neurocognitive disorders.
For HCV
- A novel first-in-class viral assembly inhibitor BIT225 targets HCV p7 protein
- In vitro studies demonstrate-
- Pan genotypic activity in HCV infectious clone assay
- Synergy with marketed drugs for the treatment of HCV
- High barrier to antiviral resistance
- Outcome of Studies in the clinic-
- Clinical antiviral efficacy against HCV genotypes 1 and 3
- BIT225 treatment is associated with faster viral clearance in treated HCV patients
- In phase I and II clinical trials, substantial safety, tolerability and pharmacokinetic data were observed based on three months dosing duration.
Stock Performance
The companyâs stock settled at $0.077 with a daily volume of ~1.26 million and a market capitalisation of approximately $44.11 million, on 22 October 2019. The stock has delivered a negative return of 35.65% on a YTD basis and 16.85% in the last six months.
Micro-X Limited (ASX:MX1)
ASX listed health care player, Micro-X (ASX:MX1) is engaged in the designing and manufacturing of products like mobile x-ray imaging systems for health care and security markets, which are based on the proprietary cold cathode, carbon nanotube emitter technology. The company is headquartered in South Australia and has its core R&D, engineering and production capability at its facility in Adelaide.
Carestream DRX Revolution Nano is the first product launched by Micro-X, which is an ultra-lightweight digital medical x-ray system for the rapidly expanding mobile x-ray market in hospitals and healthcare sector.
Patent for Micro-X Carbon Nanotube Emitter
On 21 October 2019, the company announced that its patent application for âLarge Scale Stable Field Emitter for High Current Applicationsâ was published under International Publication No. WO 2019/191801. This is a great achievement and key addition to the companyâs intellectual property portfolio.
The patent mentions the main elements of the carbon nanotube emitters, which are a core component of the companyâs next generation x-ray tube devices, the core technology behind all Micro-X products. This innovation is based on the essential concepts for nano-structure field emitters; hence, it provides Micro-X with a substantial barrier to competing field emitters being commercialised.
Official Opening by South Australian Premier
As per a company announcement dated 12 September 2019, The Premier of South Australia, The Hon Steven Marshall, MP was due to open a new extension to the manufacturing facility on 17 September 2019 and witness the first DRX Revolution Nano to be fitted with Micro-Xâs new, in-house designed and manufactured CNT X-ray tube, come off the production line.
UK Government Follow-On Airport Security Contract
On 5 September 2019, Micro-X announced to have secured a follow-on contract by the United Kingdom Governmentâs Department for Transport under its Future Aviation Security Solutions (FASS) program. This program is funded by the UK government and will seek to extend the performance of Micro-Xâs unique X-ray airport security imaging solution developed under an earlier contract.
Financial Information for 12-Month ended 30 June 2019
Net loss for the Group and its subsidiaries for FY2019 after providing for income tax amounted to $9.834 million compared with a loss in the previous year of $16.595 million; comprised of-
- A total expense of $4.8 million on research and development activity.
- An amount of $5.1 million was spent on employee and direct costs during the year.
There is a reduction in the loss for FY2019, and this loss was due to reductions in project development costs of $2.6 million.
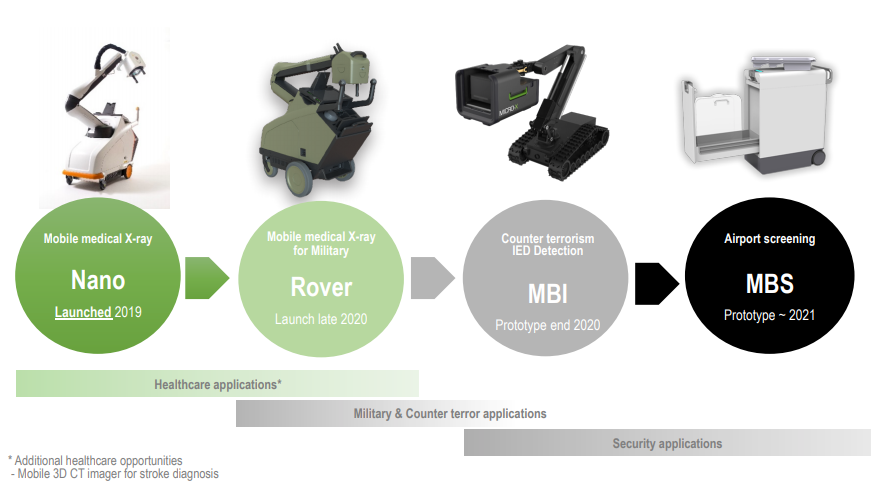
Micro-X launched its Nano product in 2019 and has a planned roadmap for the next two years.
Product Evaluation (Source: Companyâs Presentation)
Stock Information
The stock of MX1 closed the dayâs trading at $0.310 on 22 October 2019, going down by 3.125% with a daily volume of nearly 103,165 and a market capitalisation of approximately $165.02 million. The stock has a 52 weeks high price of $0.415 and a 52 weeks low price of $0.195.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.