Amid the unprecedented market instability due to the COVID-19 pandemic, investors are keenly focusing on investing in lucrative shares having the highest value in the market. The coronavirus outbreak has changed the dimensions for the businesses across the globe. While several companies are struggling with declining demand and their operations coming to a standstill, the COVID-19 crisis has created an opportunity for the healthcare sector. And the endless requirement for healthcare products and services make this sector one of the most popular options when looking for a safe bet.
DO READ: Unveiling Current Challenges & Opportunities in Healthcare Space Amidst Virus Threat
On Friday, 22 May 2020), the S&P/ASX 200 healthcare sector index settled at 41,788.0, indicating a fall of 2.04% compared to its previous close, while benchmark index S&P/ASX 200 traded at 5,497.0, a drop of 0.96%. However, two ASX-listed healthcare stocks (Antisense and Invex) ended the week with a surge in their stock price.
Both the healthcare companies have reported favourable clinical trials outcomes that are well received by investors, as reflected in the share price movement. With this positive news towards the end of the week, the management, and the stakeholders of the two players might be having a good, relaxing weekend.
Let us discuss these two ASX-listed healthcare stocks in detail.
Antisense Therapeutics Limited (ASX:ANP)
On 22 May 2020, stock of ANP soared by 39.583% to close at $0.067. The Company has a market cap of $23.46 million and ~488.79 million shares outstanding. The stock’s 52-weeks high and low price stood at $0.145 and $0.029, respectively.
About the Company:
Biopharmaceutical company Antisense Therapeutics Limited is engaged in the development and commercialisation of antisense therapies for large markets with high unmet need. The products of Antisense are in-licensed from Ionis Pharmaceuticals Inc, a world leader in antisense drug development and commercialisation.
The drug pipeline of Antisense comprises two drug candidates: ATL1103 and ATL1102. Both these candidates are under clinical development, ATL1103 for the treatment of acromegaly and ATL1102 for multiple sclerosis and Duchenne Muscular Dystrophy (DMD).
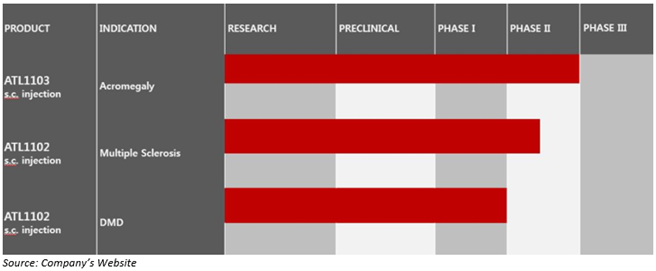
Antisense ATL1102 Final Phase II trial Reported Strong Initial Efficacy for DMD
According to an ASX announcement dated 21 May 2020, the Company disclosed that the Phase 2 clinical study of its immunomodulatory therapy, ATL1102 for DMD had met its primary endpoint confirming the safety and tolerability of ATL1102 for advancement into a potentially pivotal Phase 2b clinical trial.
Notably, the final clinical trial outcomes have also proved the positive effects of ATL1102 on the secondary trial endpoints that assessed its efficacy and activity.
Also, measurement of the participants functional capacity and effects on immune cell numbers in the blood was confirmed in this trial.
Additionally, the Company is gratified to report that magnetic resonance imaging (MRI) assessment of the upper limb muscles of the DMD patients has also demonstrated beneficial outcomes of the drug in stabilising the percentage of a fat fraction within the forearm muscles.
Now, what is next for Antisense?
Antisense highlighted that the Company is delighted by the phase 2 trial outcomes and with the assistance of international experts which offers support to start ATL1102 Phase 2b trial.
Moreover, the next milestone in preparation for submission of clinical trial application for a Phase IIb trial across Europe and the UK is scientific advice from European Medicines Agency (EMA) which is due in mid-2020. The preparation of submission for Orphan Drug Designation for the use of ATL1102 in DMD in underway across Europe and the US.
Parallelly, encouraged by observing the anti-inflammatory effects of ATL1102, the Company is researching on finding treatment for other potential diseases including ones that are improperly regulated by using corticosteroids, for example, neurological conditions and muscular dystrophies.
Mark Diamond, CEO of Antisense Therapeutics, commented:

DO READ: How Pharma & Healthcare Sectors are Supporting Economic Activities Across the Globe
The stock of IXC closed the market at $1.665 up by 11.371% on 22 May 2020. The Company has a market cap of $82.23 million and nearly 55 million shares trading on ASX. The stock has delivered a positive return of 48.00% in the last three months and 134.51% in the last six months.
About the Company
Australian biopharmaceutical company Invex Therapeutics Ltd is focused on the repurposing of already approved drug Exenatide for effective treatment of neurological conditions derived from elevated intracranial pressure, for example, Idiopathic Intracranial Hypertension (IIH), traumatic brain injury and acute stroke.
Positive Trial Results for Exenatide
On 22 May 2020, Invex updated the market with the positive phase 2 clinical trial results for Exenatide. The Phase 2 trial was a randomised, double-blinded, placebo-controlled clinical trial, conducted in 16 females aged between 18-60 years having confirmed Idiopathic Intracranial Hypertension (IIH).
- The Primary endpoint of the study demonstrated Exenatide Reduces intracranial pressure (ICP) in IIH patients.
- The secondary endpoint of the study showed Exenatide was significantly beneficial in reducing Monthly Headache Days in patients treated with Exenatide.
Strength of the results for both primary as well as key clinical endpoints indicate a clear & strong drug effect in the IIH population. The results encourage progression to a single Phase 3 clinical study for registration in the US and Europe.
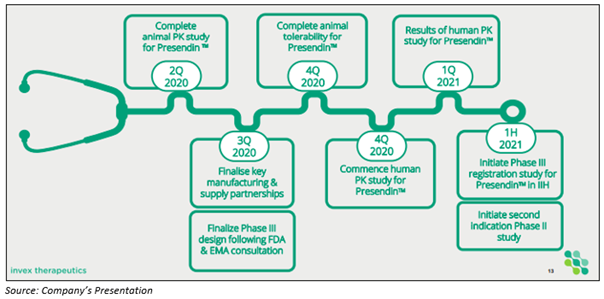
The Company also disclosed the timeline for its upcoming key milestones to market entry:
After the positive outcomes from the Exenatide trial, Invex tapped investors for $26.2 Million at a per-share price of $1.30. This offer price represents a 13% discount to its last closing price of $1.495 and a 4% discount to the 15 days VWAP of nearly $1.35.
With the clinical trial results update the Company also provided reformulation & regulatory update, disclosing that-
- Patent applications on Presendin™ (the new formulation of Exenatide) was filed in March 2020.
- The complete animal tolerability study for Presendin™ is anticipated in the fourth quarter of 2020.
- Phase 3 clinical trial design submitted to US Food and Drug Administration and European Medicines Agency (EMA) for feedback on the overall design, endpoints, statistical plan:
- Based on Phase 2 clinical trial data, Invex has the option to use either Visual Acuity or Monthly Headache Days.
- The final design lock is planned for the third quarter of 2020.
- Invex is on track to begin the study in the first half of the calendar year 2021.
- Strong data reported from phase 2 study helped Invex to raise to fully fund Phase 3 clinical trial for Presendin™ through to top-line results in the second half of 2023.
- Moreover, the Company intend to initiate a Phase 2 clinical trial in IIH-WOP with Presendin™ in the first half of 2021.
INTERESTING READ: Prescription of Mental Health Medications Surges Amid COVID-19
NOTE: $ denotes Australian Dollars unless otherwise indicated.







