 |
BARCELONA, Spain, Sept. 18, 2024 /PRNewswire/ -- The highly anticipated 2024 European Society for Medical Oncology (ESMO) Annual Congress has taken place in Barcelona, Spain. As one of the most influential annual gatherings in oncology, this congress brings together leading cancer experts and researchers from around the globe, showcasing the latest advancements in the field and providing high-quality educational and networking opportunities for oncology professionals.
Biosyngen, an innovative biotechnology company specializing in immune cell therapies, is proud to present Phase I clinical data for its pioneering CAR-T therapy targeting Epstein-Barr virus (EBV) antigens for the treatment of nasopharyngeal carcinoma (NPC).

From left to right: Ma Yuxiang, Associate chief physician in Phase I Unit in Clinical Trial Center and PhD in Oncology, Sun Yat-sen University Cancer Center; Professor Zhao Hongyun, Director in Phase I Unit in Clinical Trial Center and Deputy director in Clinical Research, Sun Yat-sen University Cancer Center; Statham Liu, Senior R&D Scientist, Biosyngen
Abstract Title
Safety and efficacy of a novel CAR-T cell therapy (BRG01) targeting the Epstein-Barr Virus envelope glycoprotein in advanced metastatic nasopharyngeal cancer (NPC) patients
Abstract No.:
899P
The presentation highlights that the Phase I clinical trial of BRG01 has successfully completed dose-limiting toxicity (DLT) observations and efficacy evaluations in nine patients, all diagnosed with advanced NPC. Notably, 100% of participants had previously undergone platinum-based chemotherapy and failed at least one immune checkpoint inhibitor, including PD-1 antibodies, while 45% had also failed treatment with antibody-drug conjugates (ADCs).
Preliminary data indicates that BRG01 demonstrates exceptional safety and preliminary efficacy. In terms of safety, no cases of dose-limiting toxicity, neurotoxicity, grade 2 or higher cytokine release syndrome, or treatment-related death were observed. The most common grade 3 adverse events were transient leukopenia related to the lymphodepletion regimen.
In terms of efficacy, disease control and response rates exhibited a clear dose-dependent relationship. Higher doses led to more significant effects, with 75% of patients experiencing a sustained reduction in tumor burden and diminished metabolic activity, and several lesions achieving complete response (CR) with 100% tumor reduction.
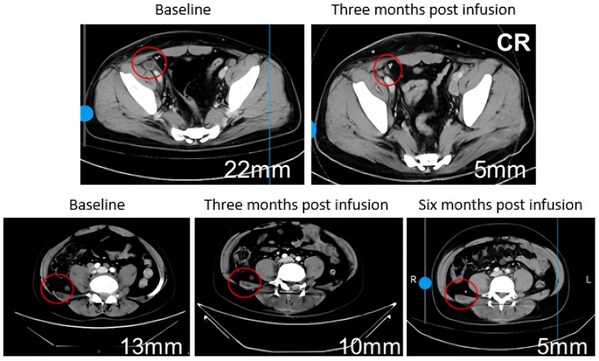
75% of patients experiencing a sustained reduction in tumor burden and diminished metabolic activity, and several lesions achieving complete response (CR) with 100% tumor reduction.
Patients treated with BRG01 have shown a progression-free survival exceeding six months post-infusion, demonstrating significantly extended antitumor effects compared to the current standard treatment, checkpoint inhibitors like PD-1 antibodies.
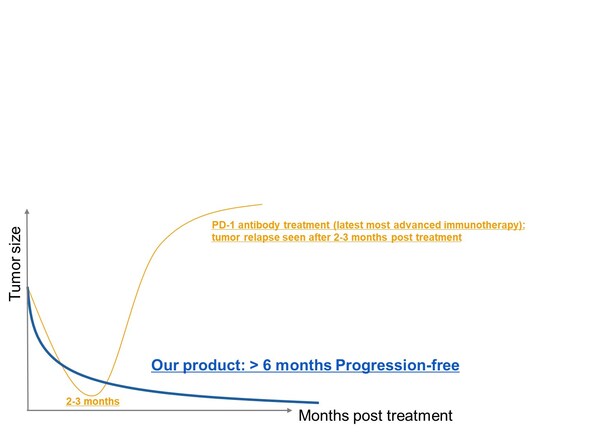
Patients treated with BRG01 have shown a progression-free survival exceeding six months post-infusion, demonstrating significantly extended antitumor effects compared to the current standard treatment, checkpoint inhibitors like PD-1 antibodies.
Additionally, BRG01 exhibited remarkable antiviral efficacy, with a significant reduction in EBV viral load in peripheral blood to normal levels post-infusion.
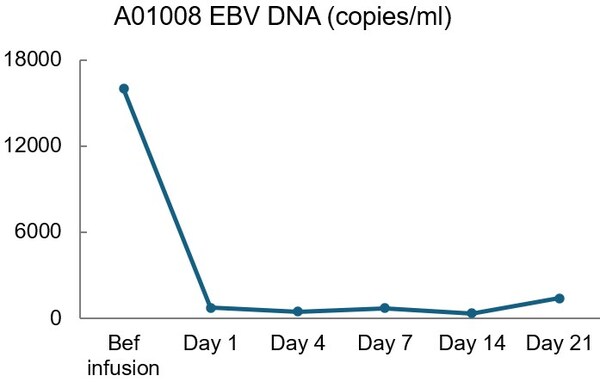
BRG01 exhibited remarkable antiviral efficacy, with a significant reduction in EBV viral load in peripheral blood to normal levels post-infusion.
Professor Zhang Li, the principal investigator (PI) of this clinical trial, and Director of the Phase I Unit at Sun Yat-sen University Cancer Center, stated, "BRG01 has the potential to be a 'first-in-class' T cell therapy for EBV-positive tumors. Its unique mechanism and robust Phase I data instill confidence in its Phase II clinical performance. We look forward to establishing its clinical efficacy through further studies, offering new hope for more patients."
As previously reported, BRG01 has received regulatory approvals for Phase II clinical trials in July (China) and August (U.S.) with patient enrollment now underway.
These data not only underscore the potential of BRG01 in treating cancer but also highlights its dual advantages in antiviral therapy, establishing a solid foundation for future clinical applications. This is likely a key factor in the Center for Drug Evaluation, NMPA and FDA's decision to advance BRG01 to Phase II trials.
BRG01 is an autologous T cell immunotherapy that expresses a chimeric receptor targeting EBV antigens through genetic modification. It represents Biosyngen's global innovation in next-generation CAR-T cell therapy specifically for EBV. The therapy received clinical trial approval from the CDE in December 2022 and the FDA in February 2023. Subsequently, it was granted orphan drug designation (ODD) and fast track designation (FTD) by the FDA in June and July 2023, respectively, marking rapid simultaneous progress in both the U.S. and China.
Biosyngen's commitment to solid tumor cell therapies extends beyond BRG01. The company is emerging as a biotech leader with a portfolio that includes CAR-T, TCR-T, and TIL therapies targeting both solid and hematologic tumors. All related product pipelines have achieved simultaneous regulatory submissions and approvals in both the U.S. and China, covering various solid tumors, including lung and liver cancer.
Regarding the initial release of BRG01's Phase I clinical data, Dr. Michelle Chen, co-founder and CEO of Biosyngen, stated, "BRG01 is the result of years of hard work by our team and a testament to the recognition of our core technology and research capabilities. Biosyngen is committed to developing innovative drugs that address unmet clinical needs in oncology. In the future, we will continue to increase our R&D investments and expedite the clinical trial and commercialization processes for BRG01, providing more effective and accessible treatment options for patients worldwide."
With Biosyngen's efficient execution and rapid research advancements, we have reason to be optimistic about achieving further clinical breakthroughs in solid tumor cell therapies in a shorter timeframe, bringing new hope to patients.



