Summary
- Small-cap healthcare companies are focused on research and development activities, concentrating efforts towards clinical development.
- Oncology-focused PharmAust recently revealed that its lead compound Monepantel (MPL) could suppress COVID-19 infection.
- Opthea disclosed positive results from the Phase 2a diabetic macular edema (DME) trial.
- DMX-200 lead compound of Dimerix has been chosen for inclusion in the protocol as a new treatment in the global clinical study program.
- Impression Healthcare commenced animal studies to evaluate IHL-675A against ARDS.
In the healthcare world, the general trend has been that smaller biotech players work on the innovation angle with research and development activities, while the more prominent players handle the marketing and commercialisation activities as they have the financial resources to handle the large-scale distribution of products.
Out-licensing deals have become a norm as they are a win-win situation for both the parties involved. While the out-licenser (small biotech players) get to receive an upfront payment along with potential milestone payments and royalties on future sales, the in-licenser gets access to a low-risk potential candidate (safety and tolerability studies already handled by out-licenser) and can expedite the process of getting a drug into the market. However, some small players willing to continue the journey of drug development and commercialisation on their own without any commercial partnerships.
Several clinical-stage companies are actively engaged in conducting studies to assess potential candidates targeting different therapy areas. These companies are focused on developing innovative products that can address the ‘unmet need’ concerns. Amidst the unprecedented market turmoil, investors are keeping an eye on small-cap stocks, as these players are relatively unscathed and continue to focus on bringing promising products to the market, either on their own or through collaborations with bigger healthcare players.
In this article, we will discuss 4 ASX-listed small-cap healthcare players who are focused on innovation and are boosting their R&D efforts for the same - PAA, DXB, OPT, IHL.
A clinical-stage healthcare company PharmAust Limited is into the development of targeted cancer treatment for both humans and animals. The Company focusses on the repurposing already marketed medicines and therefore diminishing the risks in conjunction with developmental costs. Monepantel (MPL) is the lead candidate of PharmAust, is a safe and effective inhibitor of a key driver mTOR pathway for cancer therapy.
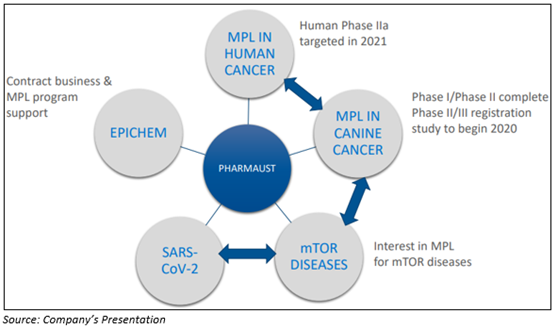
PharmAust’s Lead Cancer Candidate MPL Could Suppress COVID-19 Infection
On 4 June 2020, the Company disclosed its initial work regarding the investigation of effects of MPL and monepantel sulfone (MPLS) on novel coronavirus infected cells.
Experiments demonstrate that both infectivity and replication of novel coronavirus particles could be inhibited almost 50 to 95% in cell cultures. The minimum inhibition value from the tissue culture infectious dose assay was nearly 50%, and the maximum inhibition value was found to be 95%.
Furthermore, PAA revealed that the suppression degree for the quantitative PCR (polymerase chain reaction) individual tests was nearly 55%. PharmAust also announced that it aims to conduct further validation studies of these initial findings shortly.
It is worth mentioning that PharmAust has granted patents for the use of Monepantel in tumours & other diseases.

Stock Information
The stock of PAA was trading at AUD 0.110 on 12 June 2020 (at 02:02 PM AEST), in line with the previous close. The Company has a market cap of AUD 33.22 million and nearly 302.02 million shares outstanding.
Dimerix Limited is engaged in the development of new therapies for unmet medical needs across the worldwide markets. The Company has scalable, proprietary platform technology called as Receptor-HIT (Receptor Heteromer Investigation Technology).
The below-mentioned figure represents the clinical development of drug candidates of Dimerix:
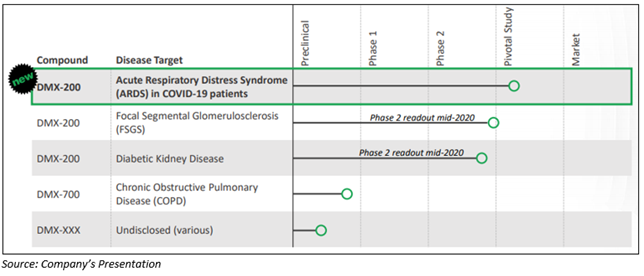
DMX-200 for Treatment of ARDS In COVID-19 Patients
On 4 June 2020, Dimerix disclosed that its lead drug molecule, DMX-200 was selected for inclusion in the protocol as a new treatment in the global clinical study program (REMAP-CAP) that is aimed for treating COVID-19 patients having ARDS. This global clinical study program is named as a Pandemic Special Study and is supported by the World Health Organization (WHO).
A global adaptive platform trial, REMAP-CAP is controlled by a network of leading institutions, specialists as well as some research groups working in partnership on an international level.
DMX-200 can offer benefits to COVID-19 patients having ARDS symptoms by lowering the inflammatory reaction in the lungs, consequently decreasing inflammation as well as subsequent fibrosis.
Notably, the Phase 2 clinical trials for DMX-200 in kidney disease remain on track and budget, with results expected in mid-2020.
Stock Information
The stock of DXB was trading at AUD 0.410 on 12 June 2020 (at 02:02 PM AEST), down by 4.651% compared to the previous close. The Company has a market cap of AUD 78.06 million and nearly 181.53 million shares outstanding.
ASX-listed biologics drug developer Opthea Limited is into developing new biologics for treating retinal disorders. Opthea is developing and advancing clinical developmental program for OPT-302 for ophthalmic diseases. The clinical development program of OPT-302 includes diabetic macular edema and wet age-related macular degeneration clinical studies.
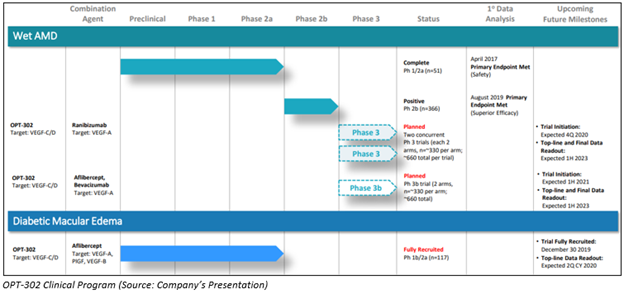
Positive Outcomes in Phase 2a Diabetic Macular Edema Trial
On 10 June 2020, Opthea disclosed positive topline findings of its Phase 2a trial evaluating the efficacy and safety of OPT-302 administered with aflibercept (Eylea®) for treatment of persistent diabetic macula edema (DME).
This trial is a randomised, sham-controlled, double-masked, proof-of-concept clinical study conducted at 53 sites in Australia, the US, Latvia, and Israel.
Moreover, Opthea disclosed that the primary endpoint of response with the combination of OPT-302 and aflibercept (Eylea®) achieved, approximately 52.8% of refractory DME patients gained five or more than five letters of visual acuity at week twelve following OPT-302 combination therapy.
Notably, the Company mentioned that OPT-302 combination therapy was well tolerated with a comparable safety profile to Eylea.
Stock Information
The stock of OPT was trading at AUD 2.380 on 12 June 2020 (at 02:02 PM AEST), down by 8.812% compared to the previous close. The Company has a market cap of AUD 702.5 million and nearly 269.16 million shares outstanding.
Impression Healthcare Limited (ASX:IHL)
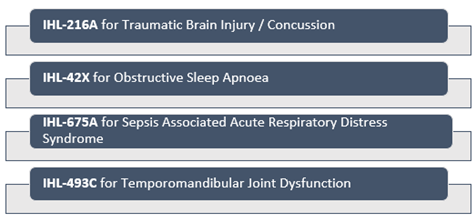
Cannabinoid medicines development company Impression Healthcare Limited is into developing unique treatment of traumatic brain injury, obstructive sleep apnoea, temporomandibular joint disorder and acute respiratory distress syndrome (ARDS).
Commencement of Animal Study for Evaluating IHL-675A Against ARDS
On 2 June 2020, Impression Healthcare disclosed the beginning of its first animal study for SAARDS (sepsis-associated acute respiratory distress syndrome) program, SAARDS is a leading cause of death due to COVID-19 as well as other infections like lung, stomach, skin, and kidney infections.
The animal study shall assess the mode of action of IHL-675A for inflammation reduction by evaluating levels of cytokine subsequent the induction of sepsis.
Moreover, the next stage of the study will consist of the investigation of specific combinations of Cannabidiol and Hydroxychloroquine, employing the learnings of the earlier stage, to examine the optimal inflammation inhibiting the response of the IHL-675A combination drug.
Notably, with this step, Impression Healthcare is assured that it will drive value for its stakeholders.
COVID-19 RELATED: Mysterious Threat of Antimicrobial Resistance Behind COVID-19
A Surge in Cannabinoid Product Sales
Impression Healthcare updated the market that the Company will discontinue the sale of oral devices from 30 June 2020 and shall entirely focus on its resources on cannabinoid sales and some other development activities.
Despite the diminished sale of oral devices, IHL foresees its revenue for the ongoing quarter to be markedly greater than the previous corresponding period due to the advantage of significantly increasing cannabinoid formulations sales.
Four cannabinoid clinical development programs of Impression are underway for the complications having no existing registered treatment options currently available:
Stock Information
The stock of IHL was trading at AUD 0.050 on 12 June 2020 (at 02:02 PM AEST), down by 7.407% compared to the previous close. The Company has a market cap of AUD 39.45 million and nearly 730.49 million shares outstanding.




