Noxopharm Limited (ASX:NOX), an Australian clinical stage biotechnology company with its focus on oncology, is known for developing a novel, potentially transformative and industry disruptive cancer drug treatment using its lead candidate, Veyonda®, in late-stage prostate cancer where no life-extending therapies currently exist. Noxopharm has made noticeable progress since its public listing in August 2016, bolstered by its novel and strategic implementation of clinical developmental programs.
Veyonda® Market Opportunity
One in five men develop prostate cancer before they turn 85. With around 14 million new global cancer cases detected every year, around 7 million cases are estimated to perhaps obtain radiotherapy benefits. In 2019, ~ 19,500 and 175,000 new cases of prostate cancer were diagnosed, and ~ 3,300 and 31,600 patients were believed to have died from prostate cancer in Australia and USA, respectively. (Source: Noxopharm Non-Deal Roadshow Corporate Presentation Jan 2020)
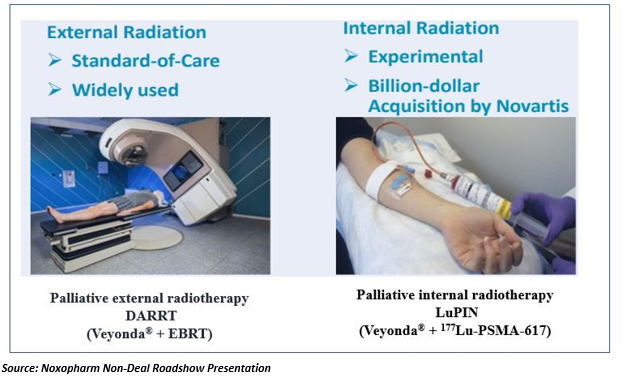
Noxopharm is targeting this potential market opportunity, seeking to commercialise a first-to-market, versatile, well-tolerated drug candidate, Veyonda®, as an adjunct to radiotherapy. The intention is to help boost the efficiency of two different kinds of radiotherapies in the treatment of late stage metastatic, castration-resistant prostate cancer (mCRPC), while aiming to restore the ability of the body’s immune system to fight and potentially halt the progress of the cancer. The two unique clinical trial designs are depicted in the figure below.
Noxopharm believes Veyonda® has future potential for use in most forms of solid cancer including as a radiation therapy enhancer and possibly an enhancer with some kinds of chemotherapy.
The company has chosen its DARRT radiation therapy enhancement program in late- stage mCRPC as the potentially quickest, lowest risk path to initial market approval. With palliative radiation treatment being the current standard of care, used generally for pain reduction with marginal impacts upon tumor burden, the management and prolonged survival of mCRPC in patients is a major unmet need which is forecast to grow in future with increasing population longevity. As per Noxopharm, the potential high demand and no known current competition in this cancer treatment space, could lead to a transformative new drug and potential blockbuster revenue.

Veyonda® in Combination with Radiation Therapy Found to be Safe and Well-Tolerated
Noxopharm recently concluded the DARRT Phase 1b study demonstrating Veyonda® + EBRT achieved a well-tolerated, durable and meaningful anticancer response at six months in a high proportion (48%) of late-stage (Stage 4) prostate cancer patients with no remaining treatment options. The final statistical outcomes are anticipated in March/April 2020.
Given the encouraging positive outcomes of DARRT-1, the company is intending to initiate a phase 2 clinical trial - DARRT-2. The protocol development is currently underway and is expecting an upsurge in overall anti-cancer effect as compared to DARRT-1 as a result of increased treatment cycles.
The medical advisory board had already been established for the DARRT-2 study with regulatory submissions anticipated in late-2020 and commencement expected in early-2021.
Veyonda® + 177Lu-PSMA-617 Underscores Noxopharm’s Confidence in Veyonda® Eventually Becoming a Standard Drug in Prostate Cancer Management
Noxopharm is also conducting an investigator-led LuPIN trial in collaboration with St Vincent’s Hospital (Sydney, Australia) led by Associate Professor Louise Emmett, to determine if Veyonda® can increase the effectiveness of a novel cancer therapy - 177Lu-PSMA-617, which is an experimental radioactive drug presently in a Phase 3 registration study (Endocyte, Inc.) likely to be completed in 2021. The drug candidate was the subject of a USD$6 billion acquisitions series by pharmaceutical player Novartis in 2018.
The interim results so far have confirmed that Veyonda® can contribute to the response to 177Lu-PSMA-617 in some trial patients, and enables more men to complete their full course of radiation therapy in a well-tolerated way, indicating that Veyonda® is safe to be used in combination with intravenous radiotherapy.
Breaking Down Frontiers- A Future Beyond Veyonda®
Noxopharm aims to go beyond palliative treatment to offer a significantly durable and well tolerated anti-cancer effect. The convincing signal that Noxopharm is pushing the boundaries with a vision to become a major drug discovery and drug development company comes from the strategic planning of new clinical programs that include the possible treatment of some brain cancers and the targeting of cancer stem cells, both of which utilise novel compounds from the company’s drug library.
Way Ahead
Noxopharm is uniquely placed at a dominant position in a critical sector, with two potential treatments for late stage mCRPC.
- Important clinical milestones ahead expected from DARRT and LuPIN trials.
- As per Noxopharm, Veyonda® is all set to generate significant revenue underpinned by progressing planning for the DARRT-2 pivotal trial.
- Approval of Veyonda® for mCRPC cancers (LuPIN & DARRT) would set in place steps to significantly expand the Company’s commercial value.
Encouraging results from Noxopharm’s clinical trials underline Noxopharm’s confidence in Veyonda® with, in what the company believes, its DARRT and LuPIN treatments to eventually become standard of care for late-stage prostate cancer, providing two new treatment options to patients and doctors.
NOX is trading at A$0.210, as on 10 February 2020 (2:41 PM AEDT).





_01_22_2025_07_55_26_045637.jpg)

