SUDA Pharmaceuticals Ltd (ASX: SUD) is a drug delivery company. It is headquartered in Perth, Australia. The company focuses on oro-mucosal administration. Currently, the company is developing low-risk oral sprays with the help of OroMist technology to re-formulate existing pharmaceuticals.
The present focus of the company is to re-formulate already existing products and make use of the regulatory and development advantages of USFDA 505(b)2 and EMEA Chapter 10(3) to reap patient benefits and ultimately shareholder returns.
Drug development across the globe is an expensive and prolonged process. Approvals for drug tests differ depending on their nature of the drug. New Drug Application and Investigational New Drug are supposed to be submitted to the FDA to obtain permission to kick start clinical testing of the new drug.
SUDA is focused on the application of its unique spray technology platform (OroMist) to existing drugs that have been approved and proven to be both safe and effective. Hence, the company is well capable to submit, what is known as an abbreviated drug approval package, known as a 505(b)(2) NDA. The 505 (b) (2) has its benefits, it allows the company to not carry out the expensive pre-clinical testing that includes animal studies for safety and toxicity. The pairing of 505(b)(2) with the companyâs OroMist technology will enable the company to create additional patent protection.
According to the companyâs portfolio update, 14 projects are classified as Investigational New Drug Status (IND) and 11 projects with clinical proof of concept or extensive clinical data. To work towards providing long-term shareholder return and deliver key projects, the company is developing Anagrelide and Midazolam oral sprays for the market. Besides these two drugs, SUD is also working on projects for partners on Sumatriptan and Cannabinoids. The funded contract around the ZolpiMist project is also being worked towards.
Apart from this, the company has kept other projects on a hold to ensure best returns of shareholders. Below are the various projects that the company has data on:
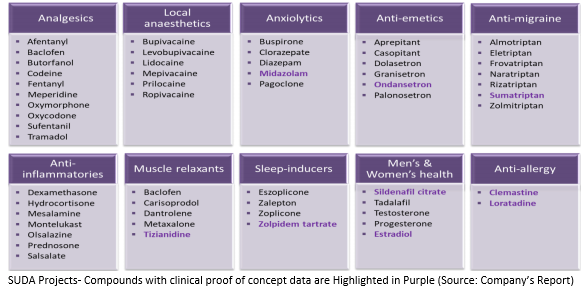
The Project Updates are as under:
- Sumatriptan Oral Spray (SUD001/SUD019)- is the worldâs most commonly used prescription medication for the treatment of migraines. SUDAâs development has proved that its 40 mg spray is a better a 100mg tablet.
- Ondansetron (SUD002)- is a medication used to prevent nausea and vomiting resulting as a side effect of chemotherapy, radiation or surgery. SUDAâs oral spray is proven to be more effective than Zofran®.
- Sildenafil for erectile dysfunction (ED) (SUD003) and Pulmonary Arterial Hypertension (PAH) (SUD004)- SUDAâs development has shown a better result with less medicine in the body to cure these.
- Midazolam (SUD005)- used for anaesthesia, procedural sedation and epilepsy, the companyâs preliminary formulation is expected to be ready for proof of concept studies in man by the 1st Quarter 2020.
- ArTiMist (SUD006)- would be updated after taking advice from the final Delegatesâ letter.
- ZolpiMist (SUD014)- is a leading hypnotic/sleep induction agent. The company aims to create ZolpiMist into a future long-term recurring revenue stream.
- Anagrelide (SUD018)- (detailed update to be provided in a few weeks)
- Cannabinoids (SUD020)- to develop various cannabis-based oral sprays, to company owns all licenses to import, store and do research.
The company has also partnered with Pfizer for the current Strides and Zelda projects. Below are the Assets/licensing deals done by SUD:
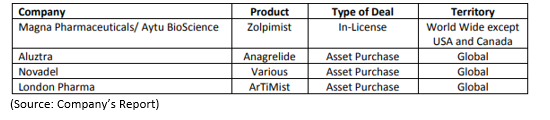
The other deals have been with Strides, Zelda, Mitsubishi Tanabe, Teva, Eddingpharm, Pfizer and Kwang Dong- making the transaction deals to be a total of 11.
The shares of SUD are trading at $0.003 on ASX (as at AEST: 1:40 PM, 14 May 2019), down by 25%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.


_06_05_2026_00_05_00_067077.jpg)



