Summary
- Imugene has attained human research ethics approval for commencement of PD1-Vaxx’ Phase 1 clinical trial in Australia.
- Sydney-based comprehensive cancer hospital, Chris O’Brien Lifehouse is the first one to attain ethics approval.
- The ethics approval validates Imugene’s completion of all necessary pre-clinical safety and efficacy testing of PD1-Vaxx needed to initiate human clinical trials.
- Imugene has planned to kick-start Phase 1 multi-centre, first-in-human, dose escalation study of PD1-Vaxx in CY20.
Australian clinical stage immuno-oncology firm, Imugene Limited (ASX:IMU) has attained a significant milestone with the receipt of ethics approval for Phase I clinical trial of anti-cancer immunotherapy, PD1-Vaxx.
PD1-Vaxx is Imugene’s B-cell cancer immunotherapy that has been designed to treat tumors like lung cancer by interfering with PD-L1/ PD-1 interaction and binding, and generate an anti-cancer effect identical to Opdivo®, Keytruda® and other immune checkpoint inhibitor (ICI) monoclonal antibodies transitioning treatment of multiple cancers.
In May this year, a ground-breaking research discovered mimotope peptides that when integrated into Imugene’s proprietary immunotherapeutic vaccine delivery platform, produce antibodies that bind specifically to immune cells expressing the PD1 biomarker.
Must Read! Get Acquainted with Imugene’s Deep Product Pipeline
Further Details on Ethics Approval
The Human Research Ethics Committee (HREC) has permitted Imugene to commence PD1-Vaxx’ Phase-1 clinical trial in Australia. Sydney-based comprehensive cancer hospital, Chris O’Brien Lifehouse is the first one to attain ethics approval for PD1-Vaxx’ clinical trial.
The Australian component of the Phase I trial will be undertaken under Australia’s Clinical Trials Notification (CTN) Scheme, signifying that the Company will inform the TGA (Therapeutic Goods Administration) of HREC approval and entire local site initiation activities.
The Company also notified that additional clinical sites will be opened in Australia, along with the US, subsequent to the US FDA (Food and Drug Administration) investigational new drug (IND).
The ethics approval validates Imugene’s completion of all necessary pre-clinical safety and efficacy testing of PD1-Vaxx needed to initiate human clinical trials.
All preclinical toxicology studies for PD1-Vaxx were completed in January this year at US-based Contract Research Organisation. Three doses were tested in the study, which delivered valuable data on the antibody generating potential of PD1-Vaxx in humans. Below are the key outcomes of the study:
- The doses were well tolerated.
- The doses generated high levels of PD1-targeting polyclonal antibodies.
- No adverse findings were recorded.
The findings of the study delivered a strong indication that Imugene’s PD1-Vaxx will generate antibodies, break tolerance in humans and may create an anticancer effect identical to other immune checkpoint inhibitor monoclonal antibodies.

PD1-Vaxx’ Phase 1 Trial to Commence Soon
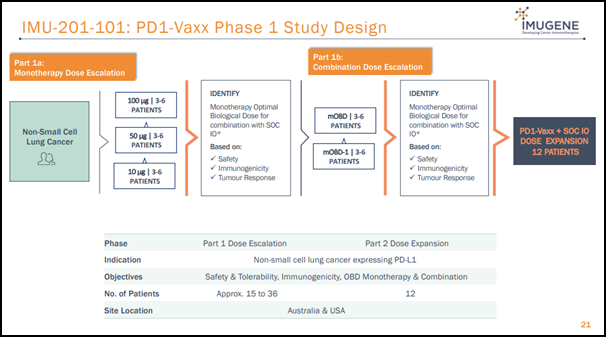
Imugene has planned to kick-start Phase 1 multi-centre, first-in-human, dose escalation study of PD1-Vaxx in CY20, targeting patients suffering from non-small cell lung cancer.
The Australian Lung Foundation has estimated that about 12,800 Australians are diagnosed with lung cancer each year. The Company will conduct PD1-Vaxx’ Phase 1 trial at up to six sites in Australia and North America under FDA IND application.
Imugene will initiate patient screening for Phase 1 trial in August this year.
Source: Company’s July 2020 Presentation
Under Phase 1 trial, medical investigators will examine three different doses of PD1-Vaxx as a monotherapy and in combination with immune checkpoint inhibitor medications.
While the key objective of the trial is to ascertain safety and an optimal biological dose as monotherapy and in combination with immune checkpoint inhibitors, the trial will also measure tolerability, efficacy and immune response.
Notably, the commencement of Australian study is a remarkable milestone for the Company and clinicians treating Australians faced with the challenge of lung cancer. According to Imugene, attainment of ethics approval speaks of the dedication and perseverance of the Company’s clinical and research team as it continues to progress on its clinical and commercial potential.
IMU last traded at $0.035, up ~2.9% on 15th July 2020 (2:56 PM AEST).







