ASX-listed clinical stage immuno-oncology firm, Imugene Limited (ASX:IMU) has released an update on the preliminary clinical development plan for the intended exclusive license of the patents encompassing the oncolytic virus technology, CF33. The company has planned to undertake two separate Phase 1 clinical trials for CF33 oncolytic virus (OV) in 2020 to test a Vaxinia construct and a CheckVacc construct.
The biotechnology company, Imugene aims to enhance the effectiveness of cancer treatments via the development of numerous novel immunotherapies that enable a cancer patientâs immune system to identify and destroy cancer cells. The company holds a strong portfolio of B cell peptide vaccines, which is expected to strengthen further with the acquisition of OV CF33.
About Oncolytic Virus CF33
Three months ago, the company announced its intention to acquire the exclusive rights of CF33 from City of Hope Cancer Centre (COH), subject to shareholdersâ approval at Extraordinary General Meeting of shareholders scheduled on 18th November 2019.
CF33 has been developed by an internationally recognised surgeon and scientist, Professor Yuman Fong, who has been recently appointed to chair Imugeneâs Oncolytic Virotherapy Scientific Advisory Board.
CF33 has been tested against a range of cancer cell lines in pre-clinical in vitro and in vivo studies. The pre-clinical data demonstrated that the CF33 virus can shrink multiple types of cancer at a low dose, and more importantly, CF33 showed a reduction in the size of both injected and non-injected tumours. CF33 is currently in the pre-clinical phase of development with the company planning to conduct Phase 1 clinical trials next year.
The oncolytic virus CF33 has been created in two different constructs:
- CheckVacc (an armed construct) with an ICI (immune checkpoint inhibitor) placed in the virus
- Vaxinia (unarmed construct).
Let us take a look at each one of these constructs in some detail below:
CheckVacc
The company notified that CF33âs Phase 1 trial commencing in 2020 will be a non-randomized, dose-escalating, open-label single-center Phase 1 study of CheckVacc, administered intratumorally in patients suffering from metastatic triple negative breast cancer (TNBC) with injectable metastatic lesions.

CF33, armed with a PD-L1 inhibitor to yield CheckVacc, has shown encouraging results in TNBC in pre-clinical studies. Considered as an aggressive form of breast cancer, TNBC impacts 20 per cent of breast cancer patients with weak prognosis upon diagnosis of metastatic diseases, largely attributable to a lack of potent targeted therapy.
Opportunities
Immune checkpoint blockade has demonstrated high hopes as a novel class of drugs for treating certain forms of advanced cancers in the past five years. The ICIs have shown efficacy in TNBCâs in the past, such as the FDA approved Genentechâs PD-L1 ICI Atezolumumab (Tecentriq) for TNBC in March 2019.
However, there are only a limited proportion of cancer patients that have attained clinical responses via ICI treatments, leaving significant opportunities for improvement possibly due to complex regulatory circuits of immune functions in deadly cancer disease.
It has been observed in Professor Fongâs preclinical studies that armed CF33/ICI combinatorial therapy can be applied to a much broader population of cancer patients.
Vaxinia
Imugene informed that CF33âs Phase 1 Mixed Advanced Solid Tumors (MAST) trial initiating next year will be a non-randomized, dose-escalating, multi-center, open-label phase 1 trial of Vaxinia administered intravenously or intratumorally in patients with solid tumours (melanoma, TNBC, bladder, lung, GI).

CF33 oncolytic virus Vaxinia has shown impressive activity in multiple forms of solid tumour in validated invivo models of colorectal, pancreatic, TNBC, lung and colon cancers. CF33 outperformed the industry leading OVâs from Genelux and Amgen in pre-clinical trials.
CF33 was found to be more effective than Amgenâs T-VEC and Geneluxâs GL-ONC1 products in killing the TNBC cell line in pre-clinical trials. When compared to T-VEC and GL-ONC1, CF33âs tumour reduction abilities in non-injected tumours was superior. While T-VEC and GL-ONC1 reduced the rate of growth in non-injected tumours compared to those mice receiving no treatment, CF33 showed a clear reduction in the size of non-injected tumours.
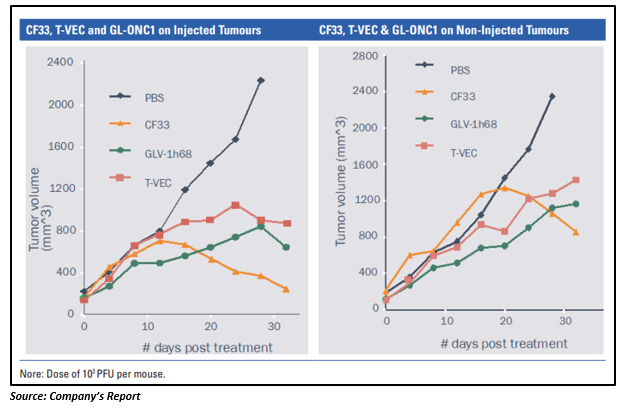
As per Imugene, Vaxinia is more powerful than its competitors, with its robust advantage being the level of dosing required, which is much lower at least in pre-clinical animal models.
Imugene has estimated a cost of USD 4.5 million for the completion of CF33 Phase 1 trials, which is anticipated to be concluded over an 18-month period.
The company believes that the addition of CF33 to its existing product portfolio would add significant value to the company in the event the clinical trials deliver encouraging results.
Stock Performance: As on 22 October 2019, IMU closed the trading session at $0.021, up 5%. The stock has delivered a return of 11.1 per cent in the last six months.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. The above article is sponsored but NOT a solicitation or recommendation to buy, sell or hold the stock of the company (or companies) under discussion. We are neither licensed nor qualified to provide investment advice through this platform.







