Immutep Limited (ASX IMM) is an ASX listed healthcare company working in biotechnology space on a global scale. The company is developing immunotherapeutic products to treat cancer and autoimmune disease.
Currently, it is working on its leading product named eftilagimod alpha, which is a soluble LAG-3Ig fusion protein and plays a vital role in the regulation of the T cell immune response. Eftilagimod alpha is used as a chemoimmunotherapy for metastatic breast cancer, in the ongoing Phase IIb clinical study.
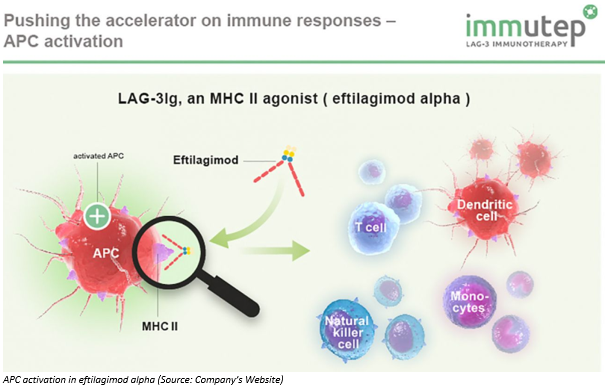
On 17th May 2019, the company announced more mature data with respect to its part B of ongoing phase I TACTI-mel clinical study of its leading product eftilagimod alpha in patients with melanoma. Dr Frédéric Triebel, Chief Scientific Officer (CSO) and Chief Medical Officer (CMO) of Immutep presented this data at the World Advanced Therapies & Regenerative Medicine Congress & Expo 2019 in London on 17th May 2019.
This multi-center, open-label clinical trial involves four cohorts of six patients to evaluate the combination of eftilagimod alpha with anti-PD-1 therapy KEYTRUDA® (pembrolizumab) with unresectable or metastatic melanoma. Different dosages of eftilagimod alpha, including 1 mg, 6 mg and 30 mg in combination with pembrolizumab, were tested with each cohort of six patients.
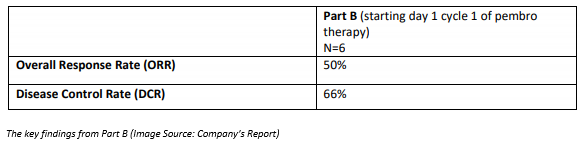
The part B of the clinical trial includes a cohort of 6 patients at 30 mg of eftilagimod alpha in combination with pembrolizumab with a treatment duration of 12 months, starting at cycle 1, day 1.
Patients in part B continue to report positive results in terms of tumour reductions after 9 months of treatment, which is in line with the previously released data for 6 months of combination treatment.
Companyâs recent and upcoming industry conference participation
On 10th May 2019, the company announced its complete schedule for the upcoming Industry Conference Participations.
From 14th â 15th May 2019, the company attended New York Academy of Sciences, Frontiers in Immunotherapy. A panel discussion was held at the conference wherein Dr Frédéric Triebel participated in the topic âNovel Approaches from Biotechâ.
From 15th -17th May 2019, the company attended another meeting in the UK (as mentioned above). Here, Frédéric Triebel discussed eftilagimod alpha, which is a soluble LAG-3Ig fusion protein.
From 31st May â 4th June 2019, the company would attend the American Society for Clinical Oncology (ASCO) Annual Meeting, wherein the company would be delivering on the topic âDevelopmental Immunotherapy and Tumor Immunobiology.â
Substantial shareholder notice
On 8th May 2019, the company announced that its initial substantial shareholder, The Bank of New York Mellon Corporation (BNYMC) now has a total voting power of 32.8%, having approximately 1.1 billion shares.
Also, on 22nd March 2019, the company announced that it has received US patent for IMP321.
Stock Performance
The market capitalisation of the company is A$98.12 million. The stock has 52-week high and low of A$0.056 and A$0.024, respectively. The stock traded and closed at an intraday high of A$0.030, as of 17th May 2019. The last one-year return of the stock is 3.5%, and the YTD return also stands at 3.5%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.