Highlights
- Prescient marked a number of developments in 2022, cementing its lead in cancer therapy innovation.
- PTX-100, being studied in a type of rare blood cancer, continued to demonstrate encouraging clinical activity with a very favourable safety profile.
- PTX-200 acute myeloid leukaemia (AML) cohort was expanded following favourable outcomes.
- Prescient launched two new cell therapy platforms in 2022 called CellPryme-M & CellPryme-A.
- The period saw new patents and successful capital raising programs.
- Read further to know the details!
2022 was another productive year for Prescient Therapeutics (ASX:PTX), which is developing personalised medicine for treating cancer. The clinical-stage oncology firm’s innovative pipeline of personalised medicine includes cell therapies and targeted therapies covering several indications.
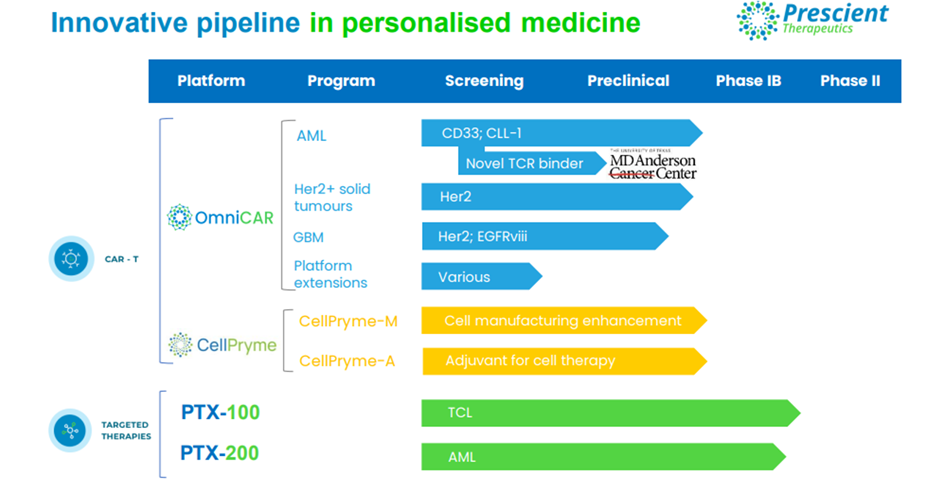
Image source: company update
Owing to its persistent efforts, Prescient this year advanced with its research work and some major developments despite sector headwinds.
PTX-100 study in rare blood cancer makes headway
In the first half of 2022, PTX initiated the enrolment in the expansion cohort of the Phase 1b trial of PTX-100.
“PTX-100 is a first-in-class prenylation inhibitor that disrupts oncogenic Ras pathways in cancer cells.”
The study in T-cell lymphomas targets the complete expansion cohort enrolment by the end of 2022. T-cell lymphomas are a group of aggressive and rare blood cancers with significant unmet clinical need.
In October, Prescient announced that PTX-100 continued to demonstrate encouraging clinical activity with a very favourable safety profile. The company also informed about plans to add more refractory cutaneous TCL (CTCL) patients to the trial.

Data source: company update
PTX-200 in Phase 1B trial with acute myeloid leukaemia patients
In May, PTX announced the expansion of cohort in the Phase 1b clinical study of PTX-200 and cytarabine in patients with relapsed and refractory acute myeloid leukaemia (AML). The cohort was expanded at 45 mg/m2 of PTX-200.
The development followed another patient with a complete remission and no dose-limiting toxicities at this dose level.
“PTX-200 is a novel PH domain inhibitor that inhibits an important tumour survival pathway known as Akt.”
CellPryme-M – a platform technology for enhancing cell therapies
2022 saw the launch of the company’s second cell therapy platform, CellPryme-M, a high-performance cell therapy manufacturing enhancement technology. CellPryme-M, developed by Prescient in collaboration with the Peter MacCallum Cancer Centre, produces superior cells during the cell manufacturing process.
As per the company, the newly launched platform also provides new ways for an entirely new business prospect to licence CellPryme-M to other cell therapy companies.
CellPryme-A - another cell therapy enhancement
During CY22, Prescient added another cell therapy enhancement to its portfolio by launching CellPryme-A.

© 2022 Kalkine Media®, Data and image source: Company update
US patent for OmniCAR
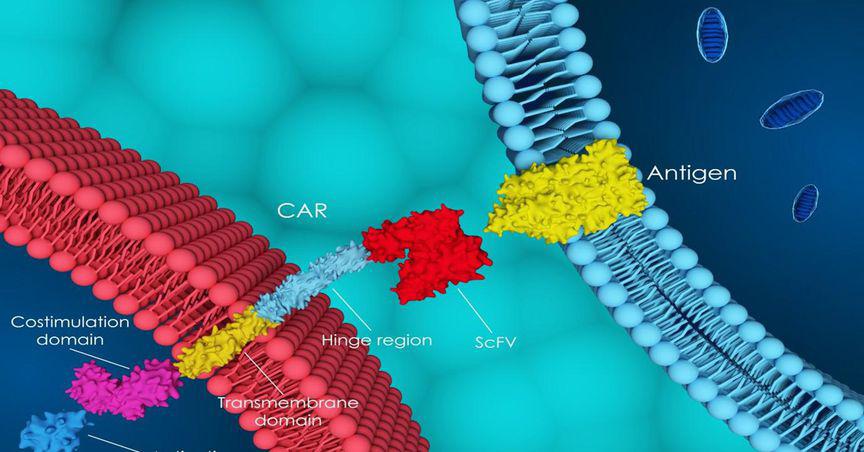
Prescient bolstered the protection of its innovative platform OmniCAR by securing a major US patent entitled “SpyCatcher and SpyTag: Universal Immune Receptor For T Cells”. The patent provides protection until at least 2039.
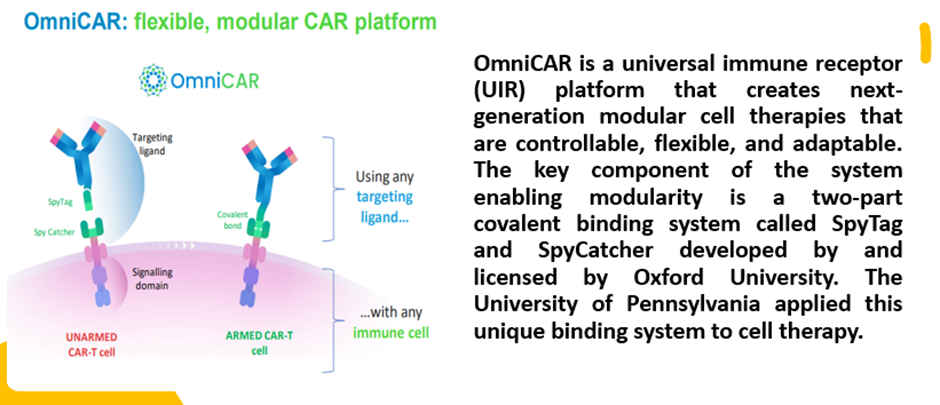
© 2022 Kalkine Media®, Data and image source: Company update
Major collaborations boost clinical developments
Prescient executed some important collaborations related to OmniCAR platform in August. Firstly, it signed a manufacturing services agreement with Q-Gen Cell Therapeutics (Q-Gen). The deal with specialist cell therapy producer was related to its OmniCAR cell lines for forthcoming clinical trials, and initial work has already started.
Secondly, with a focus on speeding up OmniCAR platform development, Prescient signed an agreement with Thermo Fisher Scientific. The pact aims to develop the next-generation OmniCAR cells that can be manufactured with unparalleled reproducibility, reduced costs, and higher efficiency.
Moreover, the company made headway in its goal towards developing best-in-class, adaptable CAR-T cell therapies targeting haematological malignancies.

Data source: company update
Financial position boost with SPP and top-up placement
The period saw Prescient undertaking capital raising programs to boost its financial position, to progress the clinical development of its targeted therapies. Further, the funds would aid in advancing the company’s cell therapies towards first-inhuman clinical studies.
The SPP exceeded its target and succeeded in raising more than AU$8.76 million.
Further, the company raised AU$2.5 million via a top-up placement. Together with SPP, the total proceeds summed up to nearly AU$11.3 million.
Prescient believes that 2023 looks to be a very promising year as it builds on this progress and approaches several transformative milestones across its targeted and cell therapy programs.
PTX shares were trading at AU$0.125 in the early hours of 22 December 2022.