Health care sector is one of the popular sectors among investors, as it is one of the high yielding sectors. However, at times, investors also lose their money, when health care stocks engaged in research & development activities fail to deliver expected outcomes from their ongoing clinical trials.
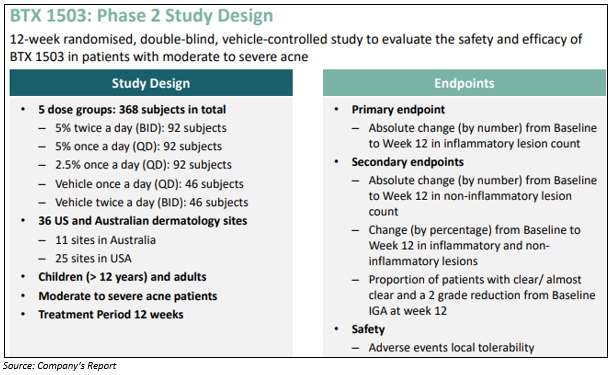
Here in this article, we have discussed Botanix Pharmaceuticals Limited, which in an ASX update on 23 October 2019, unveiled trial results for the BTX 1503 acne Phase 2 study. The stock witnessed massive selling pressure and dropped more than 50% intraday before closing the dayâs trade down by 11%. The street was disappointed with the trial results, unveiling primary endpoint of reduction in inflammatory lesions not achieving statistical significance. However, the secondary endpoint of reduction in non-inflammatory endpoints, was statistically significant.
About Botanix Pharmaceuticals Limited (ASX:BOT)
Health care sector player based in Perth (Australia) and Philadelphia (US), Botanix Pharmaceuticals Limited (ASX:BOT) is a developer of topical treatment for improving the lives of patients with severe skin conditions. The company has a license (exclusive) for Permetrexâ¢, a novel transdermal drug delivery technology for direct skin delivery of active pharmaceuticals. Botanix has a good dermatology pipeline for synthetic cannabinoids (CDBs), which are in Phase I and Phase II of clinical trials. First products are planned to enter the clinic in the second half of CY2019.
Key Results from Phase 2 Trial
- All the doses tested for BTX 1503 were found to be safe.
- There were no serious adverse reactions or treatment related discontinuations reported during the trial.
- BTX 1503 showed a positive effect on reduction of acne lesion.
- Once-daily application of BTX 1503 had the best results, which is the ideal dosing regimen from patient compliance as well as a commercial perspective.
- For non-inflammatory lesions, there was a clear separation of BTX 1503 5% once a day vs vehicle - 40.8% vs 26.4% for inflammatory and 38.1% vs 5.1%.
- BTX 1503 showed a consistent impact on inflammatory lesions in the complete study with a significant reduction in the non-inflammatory lesion.
Botanix Pharmaceuticals is now moving forward with its clinical program with a Phase 2 meeting with the Food and Drug Administration. The company is targeting to complete the enrolment for a phase 2 patient study in atopic dermatitis in the fourth quarter of CY2019, while data from this study is expected in the first quarter of 2020.
Owing to the strength and statistical significance of Australian data in addition to the overall efficacy and safety from phase 2 study delivers confidence to the company to move ahead with preparation for phase 3 clinical studies.
Stock Information
The stock of BOT closed trading at $0.105 on 24 October 2019, down 16% from its previous close, with a daily volume of ~27 million and a market capitalisation of $120.56 million. In the past six months, the stock delivered a return of 19.05%, while the year-to-date return stands at 71.23%.
Let us discuss another health care sector player, operating in the small cap space - OncoSil Medical Ltd.
About OncoSil Medical Ltd (ASX:OSL)
A leading medical device company, OncoSil Medical Ltd (ASX:OSL) is focussed on oncology research & development. The company is developing solutions to advance radiation for cancer patients, more particularly for pancreatic and liver cancer. OncoSil⢠device is the lead product of the company. OncoSilâs device is currently under clinical investigation across the globe, and the company is progressing for its commercialisation.
About OncoSilâ¢
OncoSilâ¢, which is a device made for new radiation treatment of pancreatic and liver cancer, is designed to provide more concentrated and confined beta radiation in comparison with external beam radiation. This device includes microparticles containing a phosphorous-32 beta-emitter radioisotope, which has a travel path of few millimetres in tissue.
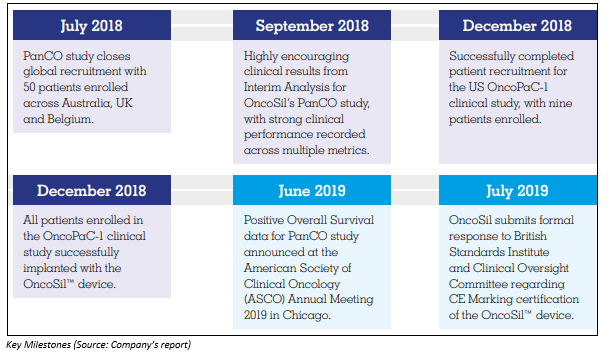
OSL Receives $3.8 Million Cash Refund
As per an ASX update on 18 October 2019, OncoSil Medical announced to have received a research & development tax incentive refund amounting to $3.8 million. The cash refund follows the submission of the tax return for financial year FY19 and is related to the expenditure on eligible research & development activities during the financial year in Australia and globally.
The amount boosted the cash balance of the company to ~ $8.5 million.
Regulatory Updates for OncoSilâ¢
In an ASX update on 11 October 2019, the company highlighted about its CE Mark review and positive regulatory developments for the US market.
OncoSil Medical is waiting for the formal assessment report from the British Standards Institute (BSI) Management after the Clinical Oversight Committee (COC) review meeting for CE Marking (required for the commercial sale of OncoSil⢠in the European Union) on 3 October 2019.
- OSL is exploring diverse US regulatory pathways for OncoSilâ¢, and as part of this, the company is investigating new indication for OncoSilâ¢.
- The company also mentioned to have recognised an excellent opportunity in the US market for cholangiocarcinoma (bile duct cancer) therapy.
- The company received a grant for Humanitarian Use Designation (HUD) from USFDA for intrahepatic and distal cholangiocarcinoma.
- CEO Mr Daniel Kenny expressed his views that OncoSil is motivated by its progress in the US and has identified a clear way to enter in the US market.
Change in Management - On 30 September 2019, the company announced the appointment of Mr Nicholas Falzon as new CFO and Company Secretary of OncoSil after Dr Ash Soman, CMO and Mr Tom Milicevic, CFO & Company Secretary, tendered their resignation.
FY2019 Highlights
- In FY2019, the company had a net cash outflow of $7.5 million from operations. The cash balance stood at $7.7 million, as the year ended on 30 June 2019.
- During the year, the company targeted a reduction in annualised cash cost base via reductions in operating expenditure and research & development expenditure. OSL also halted new clinical trial and other study activities until achieving greater certainty around CE Marking.
- As the company is working towards the commercialisation of its device and better patient outcome in pancreatic cancer, it is expecting to build its accomplishments to date throughout 2020.
The annual general meeting (AGM) of OncoSil Medical is scheduled on 29 October 2019 in Sydney, New South Wales.
Stock Information
The companyâs stock settled at $0.112, going up by 1.818% with a daily volume of 554,852 and a market capitalisation of approximately $69.38 million, on 24 October 2019. The stock has a 52 weeks high price of $0.205 and a 52 weeks low price of $0.021. In the past six months, the stock delivered a return of 107.55%, while the year-to-date return stands at -37.14%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.



_10_04_2024_16_38_50_427275.jpg)