An Australian pharmaceutical research company focussing on fibrotic and inflammatory diseases, Pharmaxis Ltd (ASX: PXS) holds a portfolio of products which are at various stages of development and approval. The product pipeline of the company is centred on its expertise in the chemistry of amine oxidase and includes a series of Lysyl Oxidase Inhibitors targeting fibrotic diseases of the liver, heart, lung and kidney, that are under clinical development.
Recently, Pharmaxis released an investor presentation on the Australian Stock Exchange (ASX), concentrated on its broad pipeline of products with multiple opportunities. Let us take a deep dive into the companyâs presentation:
Key Insights of Pharmaxis Presentation
Pharmaxis has two approved respiratory products under its clutch. Which includes:
- Bronchitol® for cystic fibrosis, and
- Aridol® for asthma diagnosis.
The company also has a proprietary and world-leading amine oxidase inhibitor chemistry platform that includes:
- AOC3 for Non- Alcoholic Steatohepatitis (NASH) and Diabetic retinopathy,
- LOXL-2 for NASH and fibrosis diseases of liver, lung, kidney and heart,
- Topical LOX for Anti-fibrotic: scarring, and
- Systemic LOX for Anti-fibrotic: Cancer.
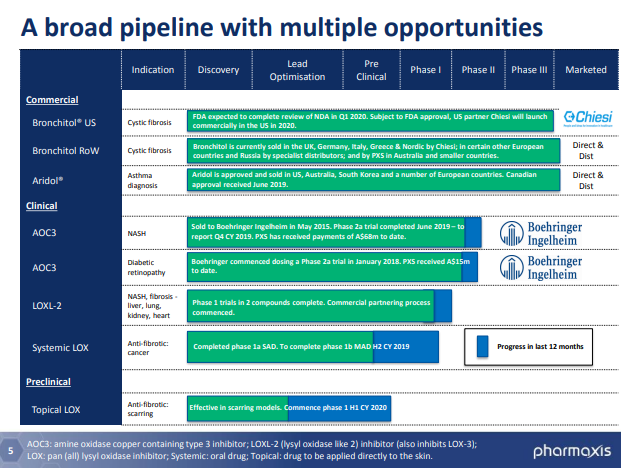
Source: Companyâs Presentation (26th July 2019)
Now, let us learn more about Pharmaxisâ products and amine oxidase platform below:
Bronchitol® & Aridol® - Mannitol Business
Bronchitol® and Aridol® are the only two respiratory products of the company that are approved in various world markets. The company manufactures and exports these approved inhaled mannitol products from its purpose-built manufacturing facility in Sydney. These products are approved by the US Food and Drug Administration, Therapeutic Goods Administration & European Union.
Bronchitol® - It is a dry powder mannitol which is used for the treatment of patients with cystic fibrosis diseases. In clinical trials, it has been observed that the product helps patientsâ clear mucus from their lungs and improve their lung function and quality of life. Bronchitol® is marketed in Europe, Russia and Australia, and is sold via a distribution network with minimal marketing expenditure.
It is expected that FDA will complete the review of Bronchitol® NDA in Q1 2020 and thereafter, the companyâs US partner, Chiesi Farmaceutici SpA, will launch the product commercially in the US in 2020.
Aridol® - It is a bronchial challenge test that is used to diagnose and manage asthma. It is approved and marketed in the US, EU, Korea and Australia via a distribution network with minimal marketing expenditure. Phamaxis also received the Canadian approval for the product in June 2019.
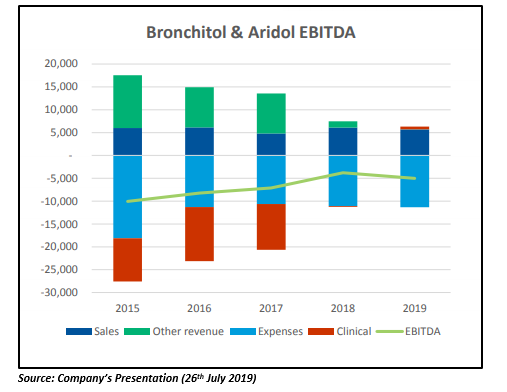
The companyâs mannitol business is expected to be profitable from 2020, driven by existing market growth plus Bronchitol® US and Russian market entry. The Russian market is poised for growth after reimbursement of Bronchitol® that was achieved on 1st January 2019. Also, Bronchitol® is expected to launch a milestone of US$10 million in H1 2020, subject to FDA approval. According to Pharmaxis, the US sales of Bronchitol®, which is likely to commence in Q3 CY 2020 and will turn the business cash flow positive.
As per Pharmaxis, the expansion of Chiesi territory in 2019 to include Greece and Nordic countries, and the appointment of EffRx as Swiss distributor in June 2019 will also work as the sales growth opportunities for Bronchitol®.
Pharmaxis expects strong sales and a robust order book for both Bronchitol® and Aridol® in FY 19. The Bronchitol® EU and Australia FY 19 in-market sales are likely to increase by over 17 per cent and 12 per cent, respectively. Further, the global sales of Aridol® are anticipated to improve by over 55 per cent in FY19. The company mentioned in the presentation that the increased sales would not materially increase the core business expenses, i.e. increasing the rate of profitability on growing sales.
AOC3
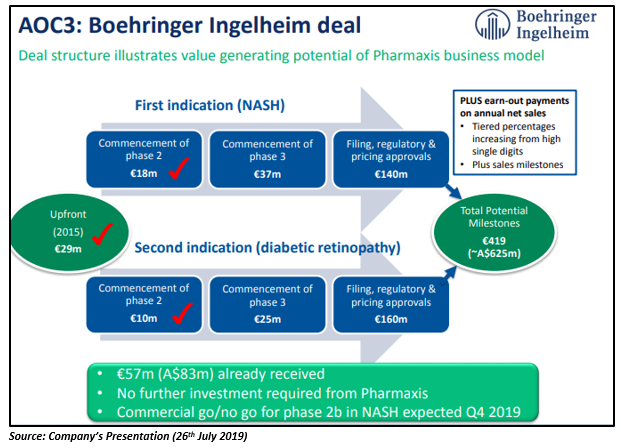
The company sold its anti-inflammatory drug candidate, AOC3 (amine oxidase copper containing type 3 inhibitor), to a leading global pharmaceutical company, Boehringer Ingelheim in May 2015, to develop it for the treatment of NASH. The deal was one of the largest in Australiaâs biotech history. As per Pharmaxis, the last patient visit under Phase IIa NASH study (including 100 patients) has been completed, and the Phase IIa clinical trial result and commercial assessment to progress to Phase IIb trial are due from Boehringer Ingelheim in Q4 2019. The company informed that it has received payments of A$68m to date from Boehringer Ingelheim under the deal.
In January last year, Boehringer Ingelheim also started dosing of patients under phase IIa trial of AOC3 in diabetic retinopathy. More than 50 per cent patients have bene recruited under Phase IIa diabetic retinopathy study in 100 patients for three months, and the clinical and commercial assessment is due from Boehringer Ingelheim in H1 2020. Pharmaxis has received payments of A$15m to date under this deal.
LOXL2
The company highlighted in the presentation that the LOXL2 inhibition program is in partnering process for NASH, IPF & other high-value fibrotic diseases, and is expected to conclude in H2 2019. During the progress and planning of the LOXL2 program, the company has been in discussions with multiple potential partners. The interest of the pharma company is driven by the search for a safe and effective inhibitor to LOXL2 and LOXL3 enzymes, safe and effective anti-fibrotic drug and drugs to complement the existing disease portfolio including lung, liver, kidney, heart, etc.
LOXL2 is one of the best oral drugs with a high-degree inhibition of LOXL2 enzyme for one full day from one dose in Phase I studies. LOXL2 is an only known drug in clinical development that also inhibits LOXL3. Pharmaxis has already completed the full analysis of the second stage of Phase I trials, and the 13-week tox studies (2 species) for both the compounds. The place of LOXL2 at the end of the fibrotic cascade provides an opportunity for Pharmaxis to treat various fibrotic diseases and use in combination with other pharma pipeline drugs.
The company also has some exciting new assets- Systemic LOX inhibitor and Topical LOX inhibitor that are advancing into the clinic phase after pre-clinical success.
Systemic LOX inhibitor
Systemic lysyl oxidase inhibition is an efficacious therapy to treat severe fibrotic indications like myelofibrosis and pancreatic cancer. The inhibition of lysyl oxidase reduces cross-linking of extracellular matrix proteins (collagen, elastin), diminishes tissue stiffness, reduce cellular stress, reduces fibrotic microenvironment and e loosens stiff stroma of primary tumours to improve access of anti-cancer drugs.
The Systemic LOX inhibitor for myelofibrosis and/or pancreatic cancer is expected to complete the Phase I trial in Q4 2019, and progress to clinical proof of concept studies in H1 2020.
Topical LOX Inhibitor
Topical pan-lysyl oxidase inhibition is a potent anti-fibrotic and anti-scarring therapeutic approach. Topical LOX inhibitor for skin scarring is expected to complete pre-clinical tox studies in Q4 2019 and progress to clinical proof of concept study in H1 2020.
The product portfolio of Pharmaxis continues to provide a pipeline of oral small molecule drugs that are targeted to treat diseases with huge markets and high unmet medical needs. The company also has a proven track record of attaining global partnerships with the world's biggest pharmaceutical companies.
Stock Performance
On 29 July 2019, the companyâs stock closed the dayâs trade flat at AUD 0.230. The stock has delivered a return of 283.33 per cent in the last five years.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.





_01_22_2025_07_55_26_045637.jpg)

