The stock-exchange listed companies that are having a market capitalisation in the range of $300 million to $2 billion are considered as small-cap stocks. The health care sector can be unpredictable for stocks of big companies, much less small-cap stocks. However, small health care companies capturing a niche have the potential to develop much faster than large-cap stocks.
The Australian share market provides various excellent opportunities for investing in top health care sector companies but there are many stocks that are not covered by analysts and have a lot of scope for the investors to generate a significant return. The investors generally consider investing in the small-cap space, since the universe of these stocks is vast.
However, before investing in any small-cap health care stock, the investors should check the following parameters-
- Product Pipeline- If there are multiple products in the pipeline and are at various stages of the clinical trial, then there is a good chance that one of them might make it to market.
- Partnerships with big pharmaceutical giants- Always check if the small-cap company has a partnership with any big names to help bring its formulations to market.
- Financials- The investors should always check how the small-cap company is performing and the current cash in hand of the company.
- What the company pledge to treat- Before investing in small-cap health care company, make sure the company has a novel, unique drug or treatment.
In this article, we are highlighting four ASX listed health care small-cap stocks and their recent developments.
Let us zoom lens for- ATH, GID, TLX, IXC:
Alterity Therapeutics Limited (ASX:ATH)
An ASX listed health care sector player Alterity Therapeutics Limited (ASX:ATH) is engaged in developing the therapies for the people having neurodegenerative diseases. The company is headquartered in Melbourne, Australia and has its offices in the United States, San Francisco, Australia and Melbourne. The company was listed on ASX in 2000 and formerly, it was known as Prana Biotechnology Limited (ASX: PBT).
The lead candidate of the company, PBT434, is the first new generation product of small molecules that are designed to reduce the pathological proteins (α-synuclein and tau proteins) aggregation implicated in neurodegeneration.
R&D tax incentive refund received
On 15 January 2020, Alterity Therapeutics updated on ASX that the company has received approximately $4.8 million cash refund under the R&D Tax Incentive Scheme of the Australian Government in relation to the cost of eligible R&D activities conducted during the FY2019.
These funds would be utilised to further R&D activities of Alterity and would add to cash reserves of the company, which was reported as nearly $10.7 million at the end of September 2019.
Orphan Drug designation to PBT434
In an ASX announcement on 14 January 2020, the company unveiled that orphan drug designation has been granted to its lead compound, PBT434 by European Commission (EC) to treat Multiple System Atrophy (MSA).
Stock Information-
On 16 January 2020, the ATH stock was trading flat at $0.021, with the market capitalisation of approximately $18.32 million. The stock has nearly 872.61 million outstanding shares in the market.
An ASX listed medical device company GI Dynamics® Inc. (ASX:GID) is engaged in developing EndoBarrier®, for the treatment of type 2 diabetes and obesity. EndoBarrier, the first endoscopically delivered medical device, is limited by federal law to investigational use only and not approved for sale. In the US, EndoBarrier is subject to an Investigational Device Exemption (IDE) by the Food and Drug Administration (FDA). GI Dynamics® was founded in 2003, and headquartered in Boston, Massachusetts.
Improvement in Cardiovascular Risk
The company revealed the positive EndoBarrier data from the Freiburg University in Germany showing improvements of biomarkers of cardiovascular risk and predicted four-year risk of major cardiovascular risk events.
Results from the study demonstrate a statistically significant improvement in various parameters such as fasting plasma glucose, HOMA score (insulin resistance), BMI, HbA1c, total cholesterol and Lp-PLA2 (cardiovascular biomarker) throughout the implant period.
Stock Information-
On 16 January 2020, the GID stock closed the day’s trade at $0.017 unchanged from the previous close, with the market capitalisation of approximately $31.11 million. Also, the stock has nearly 1.83 billion outstanding shares in the market.
Telix Pharmaceuticals Limited (ASX:TLX)
Melbourne headquartered clinical-stage biopharmaceutical company Telix Pharmaceuticals Limited (ASX:TLX) is focused on the development of therapeutic products by applying Molecularly Targeted Radiation (MTR). The company is centred towards development and commercialisation of clinical-stage oncology assets in three carcinomas- brain, kidney, and prostate cancer. Telix pharma is having its operations in the United States, Japan and Belgium.
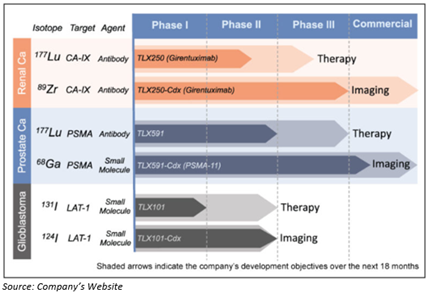
Submission of NDA clinical Briefing Package to FDA-
In December 2019, Telix Pharma revealed that the company has submitted a full clinical briefing package to the US Food and Drug Administration (FDA) for its first product TLX591-CDx1, after the receival of procedural guidance from its pre New Drug Application (NDA) meeting reported on 28th August 2019. The additional manufacturing of the product and its release analytics, as per the recommendation of the FDA, has been completed now.
The company also notified about the filing of the amended Drug Master File (DMF) with the FDA during January 2020 as part of its current new drug application submission process. However, there is no information regarding this filing of amended DMF till now.
On this announcement, Telix CEO Dr Christian Behrenbruch commented-

Stock Information-
On 16 January 2020, the TLX stock closed the day’s trade at $1.475, up by 4.24% from its previous close, with the market capitalisation of approximately $358.39 million. The stock has nearly 253.28 million outstanding shares in the market.
Invex Therapeutics Ltd (ASX:IXC)
A biopharmaceutical company Invex Therapeutics Ltd (ASX:IXC) is engaged in the repurposing of an already approved drug, Exenatide, glucagon-like peptide-1 (GLP-1) receptor agonist. The reformulated drug is used for effective treatment of neurological disorders derived from increased intracranial pressure, such as traumatic brain injury, acute stroke and Idiopathic Intracranial Hypertension.
Exenatide Phase II Clinical Trial
On 13 January 2020, Invex Therapeutics revealed that the last patient of Exenatide Phase II clinical trial had completed the twelve-week dosing schedule. This clinical trial is being conducted to treat IIH or Idiopathic Intracranial Hypertension with Exenatide.
This trial was planned to determine the significant reduction of intracranial pressure in IIH patients induced by Exenatide and to gather the necessary information to design a subsequent registration-directed, single pivotal trial to gain regulatory approval for its repurposed Exenatide in IIH in Europe and the US.
Moreover, the company anticipates the release of top-line results for this phase II trial in the late first quarter to the early second quarter of 2020.
Stock Information-
On 16 January 2020, the IXC stock closed the day’s trade at $0.985, up by 4.233% from its previous close, with the market capitalisation of approximately $51.98 million. The stock has nearly 55 million outstanding shares in the market.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.







