Summary
- COVID-19 had different impacts on different segments of the healthcare sector.
- Companies providing PPE, sanitisers, and ventilators registered growth, but demand for several other healthcare products/services reduced amid coronavirus fears.
- Sector players engaged in research & development activities continued their efforts towards developing a vaccine/treatment for COVID-19.
- Dimerix has raised AUD 5.8 million in a placement to support the global REMAP-CAP study of its lead candidate in patients with ARDS as a result of COVID-19.
- CYP has announced a key milestone to the commencement of phase 3 osteoarthritis clinical trial of CYP-004, with ethics approval and expedited regulatory pathway.
Healthcare has always been one of the attractive sectors for investments, as its growth is not much dependent on economic cycles. However, this time, the crisis is different, and different segments of the sector have witnessed different impacts, owing to the COVID-19 crisis.
Businesses that are engaged in the production of ventilators, PPE, hand sanitisers and other essential products, have registered massive growth since the beginning of the health emergency.
The other side of the story is, governments, globally, have postponed non-emergency surgeries during the pandemic period, and people have been advised to avoid any unnecessary visit to hospitals. This has hugely impacted demand for healthcare products and services.
In the wake of reduced demand, and also for regulating the healthcare system, governments were quick to implement telehealth GP consultations. The move has helped people, who were fearful of visiting doctors, to take the pieces of advice through telehealth. GP practitioners were also given guidance to offer only telehealth consultations, especially to patients with respiratory symptoms.
Last few months have been extremely challenging for the sector, as it continued to support the COVID-19 response through several activities. Australia has handled the pandemic well, and cases have almost stopped, with restrictions being eased out.
Must Read: World's Battle Against COVID-19 Seems Far From Over
Medical staff want to come back to work, but it remains to be seen how patients would re-engage with the healthcare system. It seems that in Australia, there would be no need for a second round of restrictions, and patients would soon re-engage with the health system, aiding the sector to recover quickly.
It is worth mentioning that several pharma companies are undergoing a growth phase, as worldwide pharma companies are racing against each other to develop a COVID-19 vaccine/treatment.
Must Read: Healthcare Sector Defying the Norms: COVID-19 Vaccines Under Development and Related Stocks
In that backdrop, let us discuss two healthcare stocks listed on the Australian Securities Exchange (ASX), Dimerix Limited and Cynata Therapeutics, which recently announced significant updates concerning their clinical studies.
Dimerix Limited (ASX:DXB)
Dimerix Limited (ASX:DXB) is a clinical-stage biopharma firm that develops innovative new therapies for global markets, primarily in the areas with unmet medical needs.
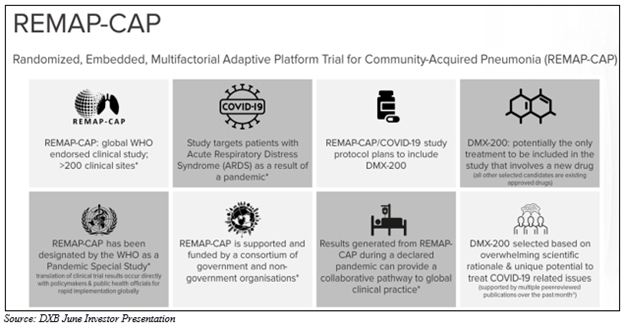
The company is currently engaged in the development of its proprietary product DMX-200. The lead candidate of the company is under investigation for the treatment of patients with Diabetic Kidney Disease and serious and rare disease Focal Segmental Glomerulosclerosis (FSGS), as well as Acute Respiratory Distress Syndrome (ARDS) in patients with COVID-19.
Dimerix Raises AUD 5.8Mn to Support Clinical Studies for ARDS in Patients with COVID-19
DXB has announced the placement of over 16.22 million fully paid ordinary shares to institutional and sophisticated investors at an offer price of AUD 0.36 per share to raise AUD 5.8 million, before costs.
The offer price of AUD 0.36 per share represents an 18.2 per cent discount to the last closing price on 17 June and a 9.4 per cent discount to the 15-day Volume Weighted Average Price (VWAP). Taylor Collison as Lead Manager and Argonaut as Co-Manager had managed the placement. Besides institutions, the placement saw strong participation and support from new and existing sophisticated and professional investors.
DXB intends to use the funds raised to support activities associated with the global REMAP-CAP study of DMX-200 in patients with ARDS as a result of COVID-19. Moreover, proceeds would be directed towards the next stage of development for the renal program covering additional manufacturing scale-up costs, logistics and distribution costs, corporate and resource costs, and general working capital.
Dimerix Chairman, Dr James Williams highlighted that it is an exciting period for the company, as two different phases 2 clinical trials into the lead candidate are nearing completion, with results expected by mid-2020.
The company is anticipating 2020 to be a transformational year for DXB with DMX-200 now entering the crucial, global study protocol in patients with ARDS due to COVID-19. Also, DMX-700 is in the process of undergoing proof-of-concept studies.
Do Read: ASX-listed Biotech Minnows- PAA, DXB Join Race for Finding COVID-19 Treatment
Dimerix CEO & Managing Director, Dr Nina Webster stated that the funds would offer flexibility in the long term for programs that include planning the success of the product in kidney and ARDS due to COVID-19 programs.
On 26 June 2020, DXB traded flat at AUD 0.355, with a market cap of AUD 64.44 million. The stock has delivered a return of 31.48 per cent and 208.70 per cent in the last one month and three months, respectively.
Cynata Therapeutics Limited (ASX: CYP)
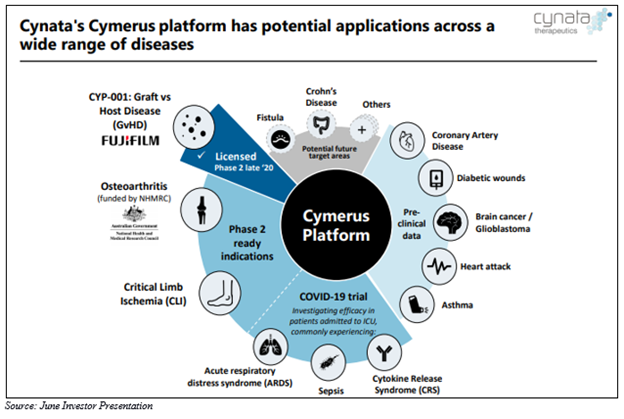
CYP is a clinical-stage biotechnology firm that specialises in cell therapeutics, with its lead product candidate being CYP-001. The company uses its proprietary CymerusTM platform technology for the development of commercially scalable cellular therapeutic products for the treatment of serious chronic disorders.
Update on Phase 3 Clinical Trial of CYP-004
The company has made significant progress towards commencing recruitment for the phase 3 osteoarthritis clinical trial of CYP-004. The trial is funded by an Australian Government National Health and Medical Research Council (NHMRC) competitive project grant and is sponsored by the Sydney University.
The University of Sydney Human Research Ethics Committee (HREC) has formally approved the phase 3 clinical trial, a key milestone to trial commencement.
Moreover, CYP has reached an agreement on an expedited regulatory pathway for the trial. The Therapeutic Goods Administration (TGA), following consultation with the university and CYP, has advised that the trial can be conducted under the Clinical Trial Notification (CTN) scheme. However, it is subject to CYP providing relevant Good Manufacturing Compliance (GMP) documentation to the trial sponsor.
The randomised, double-blind placebo-controlled trial, involving 440 patients with osteoarthritis of the knee, will take place at study centres in Sydney and Tasmania. Over a two-year period, the study aims to evaluate the effect of Cymerus MSCs compared to placebo on clinical outcomes and knee joint structure.
Cynata's Chief Operating Officer, Dr Kilian Kelly stated that the company is pleased to get the ethics approval for this significant phase 3 clinical trial in osteoarthritis patients. HREC's support is a crucial milestone achieved towards the beginning of the trial.
At the end of trading session on 26 June 2020, CYP settled at AUD 0.625, moving upward by 1.626 per cent. The company has a market cap of AUD 72.03 million.
Must Read: ASX biotech companies creating a pandemic-free buzz!





_01_22_2025_07_55_26_045637.jpg)

