Australia-headquartered clinical stage immuno-oncology player, Imugene Limited (ASX:IMU) has hit a new milestone, with the completion of GMP manufacturing and preclinical toxicology studies for its B-cell cancer immunotherapy - PD1-Vaxx.
These pre-clinical milestones have paved the way for Imugene to progress PD1-Vaxx into Phase 1 trials in 2020.

The Company is proposing to develop an anti-PD-1 immunotherapy to treat patients suffering from lung tumours that overexpress the ligand of receptor PD1 (programmed cell death 1), PD-L1/2. The hypothesis is that a polyclonal-induced B-cell antibody response will be as effective or more effective than current monoclonal antibody therapy, with improved safety.
Moreover, the therapies with monoclonal antibodies targeting PD-1 and its ligands (PD-L1 and PD-L2) relate to remarkable response rates in numerous cancers and have transformed cancer treatment.
PD1-Vaxx’s GMP Manufacturing and Preclinical Toxicology Studies Completed
Imugene notified that the FDA inspected and qualified Contract Manufacturing Organization (CMO) in the US has completed the PD-1 Vaxx’s GMP manufacturing process, including final sterile fill and finish.
Also, the final filled and finished vials of the cancer immunotherapy have finalised non-human primate (NHP) safety toxicology studies at Contract Research Organisation based in the US.
The Company mentioned that the NHP was selected for the study due to its target PD1 receptor being 100 per cent identical to human PD1. This enabled the study to provide useful data on the antibody producing potential of the cancer immunotherapy in humans.
The following outcomes were reported from the three doses tested under the study:
- Doses were well tolerated.
- No adverse findings were registered.
- Doses produced high levels of PD1-targeting polyclonal antibodies.
These findings provided a strong indication that the PD1-Vaxx will break tolerance in humans, produce antibodies, and may generate an anticancer effect similar to Opdivoâ, Keytrudaâ and other immune checkpoint inhibitor monoclonal antibodies that are revolutionising the treatment of a range of cancers.
The study was finalised under Good Laboratory Practice conditions, which is a standard needed for regulatory submissions to the US Food and Drug Administration (US FDA) in the US and the Therapeutic Goods Administration (TGA) in Australia.
Background to PD1-Vaxx
The Company acquired PD1-Vaxx from the Ohio State University (OSU) in 2018, as a part of the licensing agreement. The OSU completed the pre-clinical studies of PD1-Vaxx, which provided a proof of concept and validation for the B-cell immunotherapy.
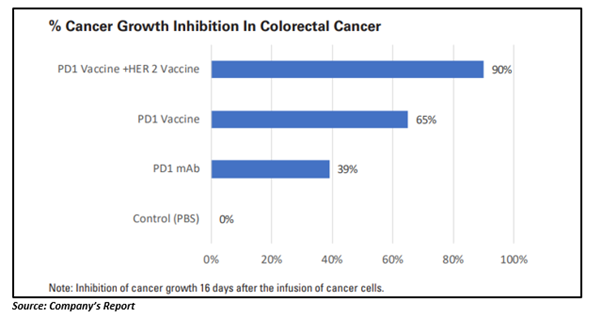
The pre-clinical study demonstrated that the immunotherapy could generate a persistent antibody response to the PD-1 protein. Moreover, the OSU undertook a study of the PD1-Vaxx in a HER2- positive colorectal cancer model, in which the immunotherapy suppressed tumour growth to a larger extent relative to an industry standard PD-1 monoclonal antibody.
One of the crucial results of the preclinical study was the tumour suppression that was discovered when the PD1-Vaxx was combined with the B-Vaxx immunotherapy, which was similar to the Company’s HER-Vaxx . Ninety per cent cancer growth suppression was realised in the colorectal cancer model, which was far better than the results from PD-1 agents on their own.
Phase 1 Trial of PD1-Vaxx to Commence in 2020
All preclinical toxicology studies completed for PD1-Vaxx allowed confirmation of clinical dose levels for the Phase 1 trial commencing in 2020. Imugene informed that the three doses tested for toxicology studies will be selected for the dose escalation stage of the Phase 1 trial.
The Company has planned to conduct a trial of the PD1- Vaxx in patients suffering from non-small cell lung cancer (NSCLC). It is the most common lung cancer type that accounts for about 80 per cent of cases.
The trial is expected to be undertaken at up to six sites in Australia and North America and under the US FDA Investigational New Drug (IND) application.

IMU closed the trading session at $0.033 on 31st January 2020.




_06_03_2026_21_29_34_162131.jpg)

