Highlights
- Imugene Limited (ASX:IMU) remains in the top three largest biotech companies in Australia today.
- The Company looks forward to achieving a range of milestones in the rest of 2022.
- With A$100 million in cash, it is one of the most financially secure biotechs on the ASX.
Australia-based Imugene Limited (ASX:IMU) has continued to make good progress and remains as strong as it has ever been in its history. In a recent letter to shareholders, the Company highlighted that it has A$100 million in cash, which makes it one of the most financially secure biotechs on the Australian Stock Exchange.
Following the release of the letter to shareholders, the Company’s shares rallied by over 13.7% to A$0.227 as of 1:52 PM AEST.
Imugene is engaged in immune-oncology therapies, with its drugs targeting over 10 disease areas. The Company has three unique platform technologies supporting six unique assets, including onCARlytics, CHECKvacc, CF33 (VAXINIA), PD1-Vaxx & HER-Vaxx. Moreover, the Company has two supply agreements with leading pharmaceutical companies and two industry/scientific collaborations.
As per Imugene, the balance of the year 2022 has some exciting developments expected for the Company. Let us take a quick look at each of these likely developments below:
HER-Vaxx
Imugene expects two Phase 2 studies of HER-Vaxx to start dosing patients in the months ahead. One study will evaluate the safety and efficacy of HER-Vaxx in combination with BAVENCIO® (avelumab), and the other with KEYTRUDA® (pembrolizumab) in patients with gastric cancer. Meanwhile, the final read-out of the HERIZON HER-Vaxx study is anticipated this year.
Recently, the Company has secured ethics approval to start Phase 2 HER-Vaxx study in Australia. The ethics approval confirms that the Company has completed all the necessary pre-clinical safety and efficacy testing of HER-Vaxx required to commence its nextHERIZON clinical study in Australia.
Related Read: Imugene wins ethics approval to commence Phase 2 HER-Vaxx study in Australia
PD1-Vaxx
In 2022, Imugene has also planned a clinical trial of PD1-Vaxx in combination with TECENTRIQ® (atezolizumab) in lung cancer patients. TECENTRIQ® is an immune checkpoint inhibitor targeting PD-L1, which has earlier demonstrated clinically meaningful benefit in several types of lung cancer, with 6 currently approved indications in the US.

In May this year, the Company signed a new clinical trial supply agreement with Roche to evaluate PD1-Vaxx in combination with TECENTRIQ® in non-small cell lung cancer patients. The Company aims to evaluate PD1-Vaxx with atezolizumab as either first-line therapy in ICI pretreated patients or ICI treatment - naïve non-small cell lung cancer patients.
Must Read: Imugene signs a new agreement for PD1-Vaxx, wins another patent for HER-Vaxx
CF33
Imugene’s oncolytic virotherapy CF33 comes in two different forms: VAXINIA and CHECKvacc, which are currently in the first phase of clinical trials.
The Company recently announced the dosing of the first patient in the Phase 1 trial of VAXINIA, which aims to recruit 100 patients across about 10 trial sites in the United States and Australia. The clinical trial is funded from existing budgets and resources and is expected to run for around 24 months.
Do Not Miss: Imugene achieves new milestone: First patient dosed in Phase 1 trial of VAXINIA
City of Hope is the first clinical institution in the United States to receive ethics approval to start the Phase 1 trial of VAXINIA. Additional clinical sites are likely to be opened across the United States in 2022.
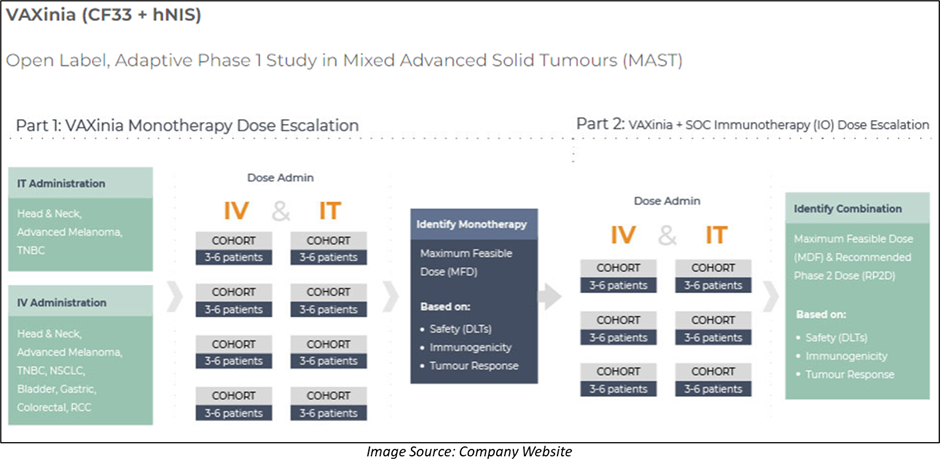
With regards to CHECKVacc, the first cohort 2 patient has already been dosed in the Phase 1 clinical trial. The current trial design will include a dose escalation, followed by an expansion to twelve patients at the final dose, which will be the recommended Phase 2 dose.
onCARlytics
Imugene informed that its oncolytic virus that expresses CD19 – onCARlytics – is awaiting publication on the combination with Celularity and Eureka CAR T therapeutics.
Last year, Imugene entered into a strategic research partnership with Eureka Therapeutics, Inc. for the treatment of solid tumours. The collaboration will explore the therapeutic potential of a combination of onCARlytics in combination with Eureka’s anti-CD19 ARTEMIS® T-cell therapy for the treatment of solid tumours.
Good Read: Imugene (ASX:IMU) targeting treatment of solid tumours via onCARlytics
With a rich pipeline and prospects, Imugene remains in the top three largest biotech companies in Australia today. The Company expects to have about 10 clinical studies supported by 5-6 FDA IND’s within the next 12 months.