Starpharma Holdings Limited (ASX:SPL) is a global lead which develops dendrimer products for pharmaceutical, life science and other applications. Located in Melbourne, its underlying technology revolves around dendrimers. SPL has two main development programs: DEP® drug delivery and VivaGel® portfolio.
 SPLâs portfolio of high-value healthcare products (Source: Company website)
SPLâs portfolio of high-value healthcare products (Source: Company website)
Dendrimers are a kind of synthetic nanoscale polymer. They are regular-sized and structured. They fit well for medicinal and pharmaceutical uses.
Today, SPL informed the exchange that it had entered a Development and Option Agreement with AstraZeneca. This deal is to progress the development of a DEP® version of an AstraZeneca major marketed oncology medicine, undisclosed as of now. With AstraZeneca, this is the companyâs second commercial agreement. The previous deal was a multiproduct licence, catering to novel oncology drug candidates.
The deal was sealed in Chicagoâs 2019 ASCO meeting and under it, the company would perform a preclinical testing of the DEP® variety of the AstraZeneca oncology product. AstraZeneca has the right to exercise its option at any point in time. Further it can licence the DEP® drug for clinical and commercial progress at any point in time. US$5m would be payable to SPL as option exercise fee, in such a case, along with industry standard progress and commercialisation indicators and escalating royalties on sales.
If AstraZeneca does not use this option, the company could license the rights for progress and commercialisation of the drug on its own or via a sub-licensee, with indicators and royalties which would be payable to AstraZeneca after the commercialisation of the end product.
The company showcased at the UBS Conference. It recorded a total revenue and income of A$0.7m in 1H 2019. The closing cash as on 31st March 2019 was A$44.7m.
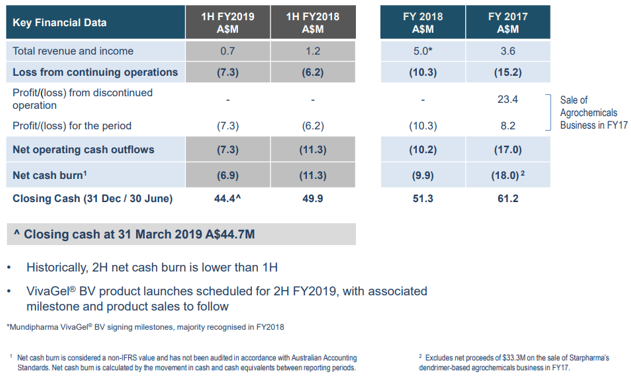
Financial position (Source: Company website)
Understanding its product portfolio, the VivaGel® BV is licensed in more than 160 countries with an on-market in Australia. Its European launch had been planned for May/June 2019. The VivaGel® condom was launched in Canada and Australia and its Japanese launch could happen in mid of 2019. DEP® is a proprietary nanoparticle drug delivery platform and by sales, it is applicable to more than 70% of the top 200 pharmaceuticals.
The company announced on 24th May that its patented nanoparticle formulation, DEP® irinotecan had showcased notable efficacy and safety benefits over the Camptosar® and Erbitux® in the irinotecan-refractory HT-29 human colon cancer model.
In their meeting held with the US FDA, the company received feedback highlighting the fact that there were various potential options to get approval as SPLâs application/NDA was inclusive of two related bacterial vaginosis indications. The FDA recognized the unmet needs regarding BV and the companyâs VivaGel® BV capability to aid towards the cause. This would be the first non-antibiotic BV treatment to treat BV.
Share Price Information:
As on 3rd June 2019, the stock traded at A$1.340, up by 2.682% from its previous trade. Its 6-month return has been a negative 16.61% whereas the YTD stands better at +23.11%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.






