Highlights
- RAD is at the forefront of advancing radiotherapy as a vital component of cancer treatment.
- Enrolment is underway for both RAD204 and RAD301 programs.
- The RAD302 program's preclinical package is expected to be completed by 2024, with a Phase 1 trial planned for early 2025.
- RAD102 is in the research & development stage, with candidate selection ongoing at Imperial College of London.
- RAD202 Phase 2 trial is planned for late 2024.
Radiopharm Theranostics Limited (ASX: RAD) has released a presentation from its recently held extraordinary general meeting, discussing its strengths, product pipeline, and upcoming plans.
RAD, an ASX-listed clinical-stage radiotherapeutics company, explores innovative products within radiotherapy, targeting integrin αVβ6, PDL-1, fatty acid synthase, HER2, B7H3, and TROP2. Leveraging insights from Antibody-Drug Conjugates (ADCs), the company utilises sophisticated targeting agents such as nanobodies.
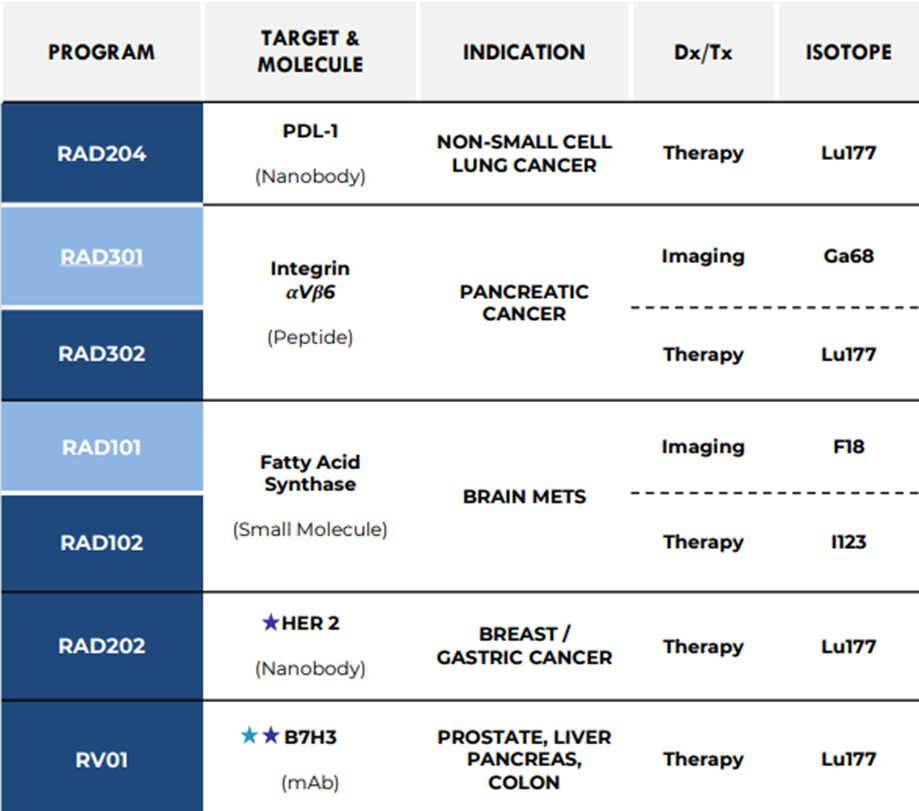
Image source: Company update
Based in Australia, the company boasts a deep pipeline of radiotherapy assets in development, including RAD204 and RAD301, currently enrolling for Phase 1 trials in Australia and the US, respectively. RAD301 has obtained orphan drug designation from the FDA.
The preclinical package of the RAD302 program is projected to conclude by 2024 with a phase 1 trial planned for early 2025.
The company is preparing an investigational new drug (IND) application for the RAD 101 Phase 2b multicentre trial in the US, targeting enrolment of 30 patients.RAD102 is in the research and development stage with candidate selection ongoing at Imperial College of London.
Preclinical and CMC stages have been finalised in the RAD202 clinical trial with a Phase 2 trial targeted in late 2024. Additionally, RAD is progressing CMC production at RV01 program, a joint venture with MD Anderson, aiming to conclude the preclinical package by late 2024.
RAD 402, linking Terbium-161 (Tb-161) to a monoclonal antibody (mAb) to target KLK3 expression, is being developed. The company is on finalising GMP CMC production. GLP Tox and biodistribution studies are expected in the second half of this year with phase 1 trial readiness anticipated in the first half of 2025.
About RAD’s team
RAD’s team comprises seasoned experts with significant expertise in both imaging and therapeutic radiopharmaceuticals. Furthermore, the firm benefits from the guidance of an extensive Scientific Advisory Board comprising accredited researchers from multinational backgrounds.
RAD shares traded at AU$0.044 apiece on 16 April 2024.







