Australian Biopharmaceutical company Exopharm Limited (ASX: EX1) is focused on developing regenerative medicine with the use of exosomes as therapeutic agents. It seeks to commercialise its proprietary product, Exomeresâ¢, earlier known as Plexarisâ¢.
In the announcement dated 22 May 2019, Exopharm reported significant progress in exosome product manufacturing and product analytics over the past three months.
Exopharm and its experienced Melbourne based Contract Research Organisation (CRO) have prepared and lodged a detailed submission to seek approval to conduct the PLEXOVAL study at a public Hospital in Melbourne.
The PLEXOVAL study will be a first in human study using exosome product made using LEAP Technology. Use of proprietary exosome product to treat humans has positioned Exopharm at the forefront in this emerging area of regenerative medicine.
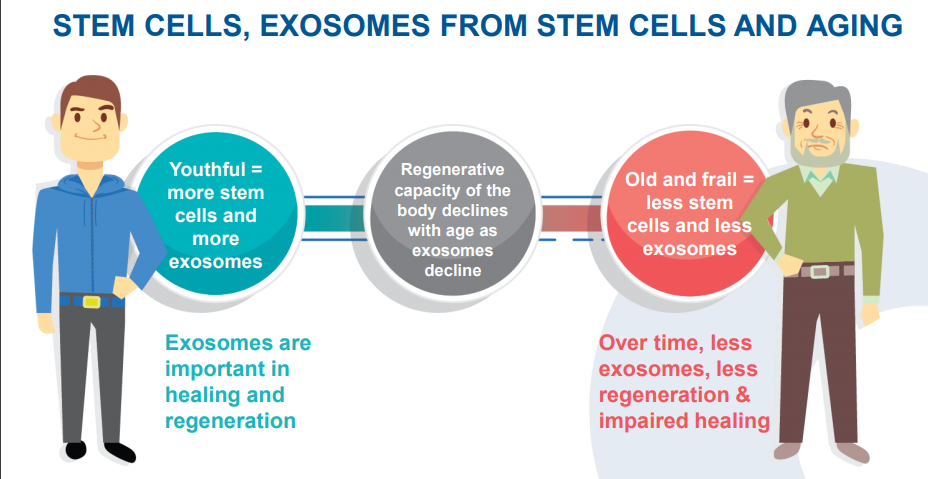 Stem Cells, Exosomes from stem cells and Aging (Source: Companyâs Investor Presentation, April 2019)
Stem Cells, Exosomes from stem cells and Aging (Source: Companyâs Investor Presentation, April 2019)
Subject to approvals, the PLEXOVAL study covers the treatment of 20 paid volunteers with autologous (from the person) Plexaris (exosome product from blood platelets) in a wound healing setting using local injection. The main readouts of the combined Phase I/IIa PLEXOVAL study will be safety, wound closure and scarring.
Exopharm anticipates the study commencing in the early second half of calendar 2019, as planned. The study is reported to go through approvals, recruitment, first dosing, last dosing, last patient follow-up and then reporting.
The companyâs core strategy is to partner its products and technologies through licenses and associated financial transactions. To support that strategy, Exopharm is progressing a wider testing program of its Plexaris and Exomere products â including non-clinical, pre-clinical and clinical testing of its exosome products for other medical conditions e.g. dry age-related macular degeneration (AMD) and osteoarthritis (OA).
With respect to companyâs non-core activities, Exopharm reported some experimental programs in process in the field of engineered exosome/extracellular vesicle (EV), due to the recent commercial interest recorded in engineered exosomes and the significant financial potential associated with it. The field of exosomes as diagnostics marks another highlight within the companyâs ongoing experimental programs.
EX1 is further accelerating development activities in areas where its LEAP Technology has likely particular potential, with the aim to add value and then seek partners for these non-core applications of the LEAP Technology.
The Ligand-based Exosome Affinity Purification (LEAP) Technology provides a proprietary technique to purify exosomes from a number of sources and for several uses. Exopharm sees its LEAP Technology as the key to solving the bottleneck problem that is delaying the production and purification of proprietary clinical grade exosomes/EVs.

EX1- Patent applied for LEAP Technology in December 2017
(Source: Companyâs Investor Presentation, April 2019)
Currently, Exopharm is pursuing commercial arrangements under which it could license partners to use the LEAP Technology in areas that do not compete with Exopharm directly. However, the discussions are at an early stage due to which outcomes remain uncertain.
EX1 last traded at $0.495, up 8.791%, as at 22 May 2019. Over the past one month, the stock has returned positive 7.06% yields, but its YTD return stands negative at -4.21%.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.







