Diversified player, BPH Energy Limited (ASX: BPH) is an Australian-headquartered firm with investments in multiple sectors, including medical cannabis, resources and biotechnology sectors. The company is commercializing a portfolio of Australian-based biomedical technologies evolving from collective research by prominent universities, hospitals and medical institutes across the country. The company offers initial stage funding, project management and commercialization strategies for a spin out company, a direct collaboration, or to obtain a license.
BPH holds investments in the following companies:
- Cortical Dynamics Ltd (4.43 per cent)
- Advent Energy Ltd (22.6 per cent)
- Molecular Discovery Systems (20 per cent)
- Patagonia Genetics Pty Ltd (10 per cent)
In an update on the ASX today, BPH informed that its major investee, Cortical Dynamics Ltd has achieved a significant Korean regulatory milestone. The company notified that Corticalâs South Korean distributor of the Brain Anaesthetic Response Monitoring (BARM) system, Globaluck has received a certificate of approval for BARM from the Korea Good Manufacturing Practice (KGMP).
BARM is a Class II medical device that has been developed by Cortical to better identify the impact of anaesthetic agents on brain activity, supporting anaesthetists in retaining patients optimally under anaesthetic.
What is KGMP certification?
Ministry of Food and Drug Safety (MFDS Korea) demands KGMP certification before placing Class II, III, and IV medical devices on the market in South Korea.
In order to get KGMP certification, the company has to submit the supporting documents to an MFDS authorized 3rd party auditor, showing that the device conforms with
- Technical;
- Risk;
- Design, and
- Associated KGMP quality system obligations.
Subsequent to submission of the KGMP certification application, the MFDS and/or a qualified 3rd party auditor (based on the deviceâs classification) inspects the manufacturing site.
An independent Korean audit of Corticalâs facility was conducted in Scoresby, Victoria during July this year, following which, Globaluck received the KGMP certification.
Corticalâs Distribution Approach
Cortical intends to develop strong relationships with major distributors in target markets, to promote product sales and support. The company entered into two major international distribution agreements in April 2018, with the following companies:
Globaluck: An exclusive five-year distribution agreement for the distribution of BARM in South Korea.
Innomed Benelux B.V.: A five-year exclusive supply distribution agreement for the distribution of BARM in the Benelux countries.
These agreements enabled Cortical to get distribution opportunities in South Korea, Belgium, Netherlands and Luxemburg. Cortical has already achieved TGA approval for BARM in Australia and the CE mark in Europe, allowing BARM to be sold and marketed initially into the Asia-Pacific, European, the UK and the South American markets. Now, the companyâs target is to develop sales in New Zealand and Australia, along with key segments of the UK, Asian and European markets.
The company has been establishing market recognition of the BARM System by working with prime opinion leaders to create a cohort of early adopters. It has been trialling the usage of the BARM System and is planning to perform the further formal clinical trials and publish the results to further validate the utility of the system.
Currently, seven hospitals are conducting trials of the BARM System in France, Australia and New Zealand. LiDCO Ltd UK is also undertaking the BARM trial at UK-based Southampton University Hospital.
Let us now discuss the key features that make BARM stand-out amongst its competitors:
BARMâs Stand-Out Features
The BARM system is developed from theoretical knowledge of physiological factors that are liable for the production of the EEG (electroencephalogram) activity, and how the EEG is disrupted by sedative and anaesthetic agents. It measures brain electrical activity more accurately and reliably during anaesthesia.
The system exclusively produces two measures:
BARM is believed to provide a competitive advantage to Cortical in the following ways:
- The BARM System can be used with Opiods, Nitrous Oxide and Ketamine, for which older, more traditional monitors cannot be used.
- As BARM directly measures the level of consciousness and the brainâs activity, patients can come out from anaesthesia immediately after the operation is over. By using this function, patients can be operated with less anaesthetic medication, leading to fewer anaesthesia associated post-operative side effects.
- BARMâs underlying algorithm is based on a model of brain electrical activity obtained from physiological standards, while other anaesthesia monitoring systems in the market make use of statistically based empirical approaches.
- BARM gives a reading within three seconds of initiation and an updated reading every second afterwards, relative to thirty seconds for a leading industry competitor; which offers for an accurate and more responsive measurement for the anaesthetist.
- It is considered to be the only EEG based depth of anaesthesia monitor to assess both analgesic (pain relief) and hypnotic (level of consciousness) state.
- BARM holds an ability to indicate a fall in awareness or sedation in fully paralysed patients.
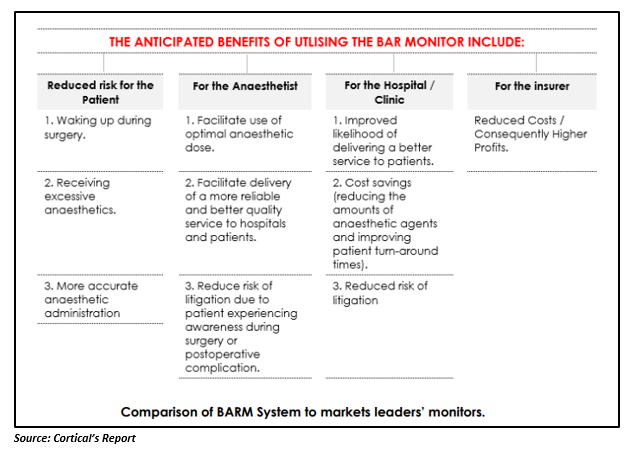
One can deduce that Corticalâs BARM enjoys a significant competitive advantage in the global market, backed by its exceptional features. BPHâs key investee is likely to create substantial value in the future, owing to its remarkable strategy and superlative product.
Stock Performance: On 30th September 2019, BPH closed the dayâs trade at $0.002. The stock has delivered a YTD return of 100 per cent, and a 1-year return of 166.7% per cent.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. The above article is sponsored but NOT a solicitation or recommendation to buy, sell or hold the stock of the company (or companies) under discussion. We are neither licensed nor qualified to provide investment advice through this platform.








