Prescient Therapeutics Limited (ASX: PTX), an Australian based clinical-stage Oncology company, dedicated towards the treatment of an array of challenging cancers with significant unmet need has recently reported operating highlights and financial results for the December quarter 2019.

Prescient’s December 2019 Qtr Financial Update
The Company held A$9.2 million cash reserves which included $1.6 million cash rebate under the Federal Government’s Research & Development (R&D) Tax Incentive Scheme.
Given the present challenge of strong US dollar versus the Australian dollar, the Company continues to manage costs wisely, in particular, the expenditure associated with the clinical trials that are being carried out in the US. To address this, Prescient further informed that, whilst it will continue to progress its product pipeline, Prescient would also seek to move trials to Australia, and/or investigator funded studies supported by non-dilutive funding.
Prescient’s significant clinical development with PTX-100 and PTX-200
- First Patient Dosed with First-in-Class Drug PTX-100 in Phase 1b Clinical Trial
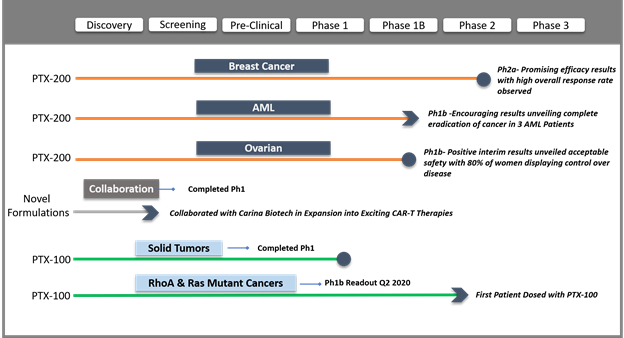
One of the key developments was the commencement of treatment in the 1st patient with its first-in-class, Ras pathway inhibitor, PTX-100 in Phase 1b clinical trial, which is currently being conducted under the supervision of world-leading oncologist Professor Miles H. Prince AM.
The study aims at establishing safety, optimal dosage as well as treatment schedule of PTX-100 for the treatment of numerous cancers where the Ras and RhoA mutations are prevalent.
Expansion of Phase 1b trial of PTX-200 in AML Patients Post Encouraging Results
During the December quarter, Prescient also announced PTX-200 Phase 1b trial expansion in patients with relapsed and refractory acute myeloid leukemia (AML) after observing promising outcomes in 3 of 15 subjects revealing total eradication of the disease in this difficult to treat patient population.
The expansion study would aim to identify the optimal PTX-200 dosing schedule with chemotherapy, minimising the side effects based on these findings.
- Promising Interim Data from PTX-200 Phase 1b Ovarian Cancer Trial
Another key accomplishment during the quarter was the positive interim outcomes Prescient’s PTX-200 phase 1b trial carried out in women detected with recurrent or persistent platinum resistant ovarian cancer demonstrating acceptable safety wherein 80% of women exhibiting disease control (being a stable disease or partial response).
· Encouraging Efficacy Findings from PTX-200 Phase 2a trial in HER2-Negative Breast Cancer
Prescient’s yet another achievement was the encouraging data from the Phase 2a trial of PTX-200 in eleven women diagnosed with locally advanced, human epidermal growth factor receptor 2 or HER-2 negative breast cancer, revealing an overall 91 % response rate.
The study showed that 2 women displayed a complete eradication of disease. One patient who also achieved a complete response post PTX-200 and paclitaxel passed away before surgery. It was further informed that till date, 9 of 10 patients being examined remain free of cancer, along with positive ongoing durability of response.
Altogether, Prescient’s strong clinical advancement underscores initial clinical proof of concept of PTX-200 with its potential as a novel Akt inhibitor.
Snapshot of the clinical developments made by Prescient is depicted in below.
- Prescient’s New Foray - Expanding Footprints into New Personalised Cancer Therapies
Prescient collaborated with a private Australian cancer research company, Adelaide-based Carina Biotech, a recognised leader in the development of CAR-T (Chimeric Antigen Receptor T cell) treatments for solid tumours. The collaboration aims to bring together Prescient’s targeted therapies expertise with Carina’s promising CAR-T technology to improve CAR-T approaches. The collaboration also broadens Prescient’s commitment to personalised cancer treatment into new cellular therapies in addition to augmenting the potential and risk profile of its clinical pipeline.
Prescient further informed that the collaborative research to be done within its current budget & does not need any additional capital, and any resulting intellectual property will be shared by both the companies.
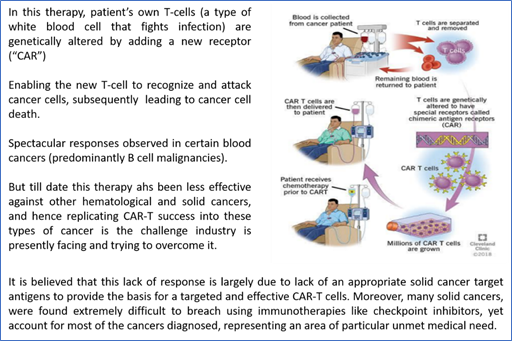
CAR-T therapies has been used to outstanding effect against some blood cancers and have drawn global investors and medical attention.
Cash Flow Report for December 2019 Quarter
PTX reported A$528,000 of operating cash inflow. Further, the Company anticipates an operating cash outflow of A$2,351,000 in the March 2019 Quarter.
In a nutshell, Prescient and its pipeline of clinical programs hold a significant place in the key developments being made in precision oncology at a global scale and pledge to revolutionise cancer treatment. Prescient’s clinical progress is backed by a strong and efficient board and management team having a focus on delivering the clinical milestones and creating significant long-term value for stockholders.
PTX was trading at $0.052 on 3 February 2020 (AEDT: 1:10PM).
To know more about Prescient Therapeutics Limited, click here.
To stay updated with PTX company activities and announcements, please update your details on their investor centre.







