Australia-based clinical stage immuno-oncology firm, Imugene Limited (ASX: IMU) is involved in the development of various new and novel immunotherapies that attempt to stimulate the patientâs immune system to eradicate and treat tumours. Imugeneâs unique platform technologies seek to harness the patientâs immune system against tumours, potentially attaining an alike or larger effect than artificially produced monoclonal antibody (Ab) and other immunotherapies.
The companyâs robust product pipeline includes numerous B cell peptide vaccines including HER-Vaxx (anti-HER-2), PD1-Vaxx (anti-PD-1), B-Vaxx (anti-HER-2), a combination of B-Vaxx and PD1-Vaxx along with an oncolytic virotherapy (CF33). The company intends to treat multiple cancers in combination with evolving immunotherapies and standard of care medications.
In a recent update on the ASX, the company notified about the appointment of Professor Yuman Fong MD, the Sangiacomo Family Chair in Surgical Oncology and Chair of the City of Hope Department of Surgery, to lead its recently formed oncolytic virotherapy Scientific Advisory Board.
Oncolytic Scientific Advisory Board
As per Imugene, the creation of Oncolytic Scientific Advisory Board depends upon the companyâs proposed licensing transaction of Oncolytic Virus CF33 from City of Hope, which is contingent upon the shareholder approval at the Extraordinary General Meeting (EGM) scheduled for 18th November 2019, and a number of customary conditions precedent.
In July 2019, Imugene informed about its intention to obtain an exclusive licence to CF33 from the Los Angeles-based City of Hope Cancer Centre (COH), conditional on shareholder approval.
Under the conditions of the licence agreement, Imugene was to acquire the exclusive rights to commercialise and develop CF33 oncolytic virus, for which the company agreed to pay COH licence fees to include the following-
- annual maintenance fees creditable against future royalty payments,
- upfront fee,
- performance based consideration linked to the achievement of certain milestones and commercial outcomes,
- net sales based royalty payments, and
- sub-licencing fees.
CF33 was developed by Professor Yuman Fong MD at COH in Los Angeles, whose clinical work has focused on oncolytic virotherapy for more than 30 years. He is a physician, surgeon and a renowned cancer researcher of the world. Dr. Fong is a globally recognized expert in pancreatic and liver cancer, who has developed many new surgical techniques and instruments.
Why Oncolytic Virotherapy?
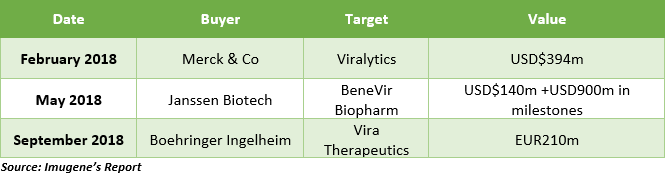
Oncolytic Virotherapy (OV) is drawing significant attention of great pharma players like Boehringer Ingelheim, Janssen and Merck, who have made three acquisitions totalling over $1.0 billion in 2018 alone, including the acquisition of Viralytics which was formerly listed on the ASX.
Oncolytic viruses are an emerging form of immunotherapy with the development led by advancements in genetic engineering. As per a body of clinical research, the use of oncolytic viruses when combined with checkpoint inhibitors holds the potential to enhance the efficiency of cancer treatment without raising toxicity.
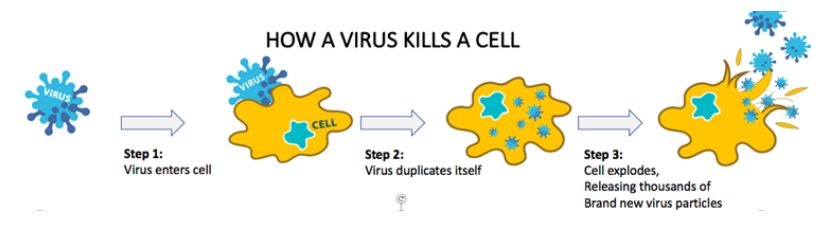
Source: Companyâs Report
OVs are developed to both activate the immune system against cancer cells and kill tumour cells, with the potential to improve survival and clinical response. They hold the potential to transform oncology by providing a potent payload in a targeted fashion that stimulates the immune system and by directly inducing tumour cell death.
Imugene believes that the addition of oncolytic virus CF33 to its product portfolio has the potential to add considerable value to the company if clinical trials deliver positive results.
If the acquisition gets approved by shareholders at the EGM, the company will commence a Phase I clinical trial for CF33 in 2020. In Phase 1 trials, the company intends to assess the safety and efficacy of CF33 across a range of advanced solid cancers including gastric, lung, bladder, triple negative breast (TNBC) and melanoma cancers.
Imugene expects to complete the Phase I trials over an 18-month period, which is likely to cost USD 4.5 million to the company. The company believes that if CF33 successfully concludes the Phase I trials and commences Phase II trials, there might be a number of suitors to acquire the licence of the oncolytic virus given the strong industry interest in oncolytic virotherapies.
As per Imugene, the acquisition of CF33 would possibly pull interest from big pharma players, backed by the research that underlines the therapeutic advantage of oncolytic viruses when coupled with additional immunotherapies.

Together with leading specialists, international cancer experts and medical professionals, Imugene believes that its immuno-oncology therapies will become foundation treatments for cancer.
Stock Performance: As on 21 October 2019, the companyâs stock is trading at $0.021 (as at 1:38 PM AEST). The stock has delivered a decent return of 46.67 per cent in the last three months.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. The above article is sponsored but NOT a solicitation or recommendation to buy, sell or hold the stock of the company (or companies) under discussion. We are neither licensed nor qualified to provide investment advice through this platform.