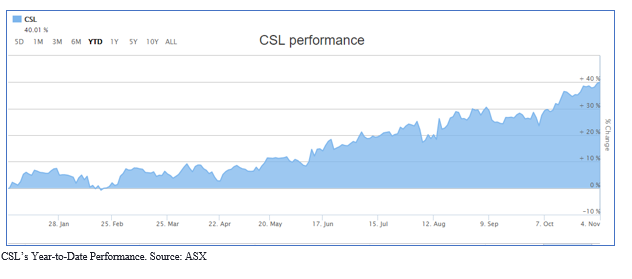
Pharmaceutical giant with one of the largest and fastest-growing protein-based biotechnology business, CSL Limited (ASX:CSL) continued to be the market darling throughout the year with its share price reaching its all-time high of $262.0 on 4 November 2019. In the last 30 days, the CSL shares gave return of 8.83% adding to what has been an outstanding year for CSL shares. This monthly upsurge in stocks could be attributable to increasing demand for immunoglobulin resulting in a shortage of supply, as reported by US FDA earlier in August 2019.
As on 6 November 2019, CSL shares were trading at $261.30, up 0.107% (at AEST 2:52 PM). The market capitalisation of the company stands at $118.35 billion and around 453.87 million outstanding shares. The 52 weeks high of the stock is noted at $263.18, with an average (year) volume of 759,119. The PE ratio is noted at 43.17x and EPS at 6.04 AUD. CSL generated an annual dividend yield of 1.02%. The most recent dividend amount was noted at 145.455 cents, ex-dividend was recorded at 10 September 2019 and dividend was paid on 11 October 2019. The total dividend was up 18% for the given period. The year-to-date investment return from CSL shares was noted at 40.66%.
2019 has been a Stellar Year for CSLÂ
An outstanding year for the CSL is reflected from its share price graph which showed a steady and gradually increasing upward trend underpinned by the robust financial performance, with a strong upsurge in revenue by 11% for FY2019 and on a constant currency basis, the revenue increased by 17%. CSL reported $1,919 million net profit after tax and US$1.85 total full-year dividend per share.
The company recorded interim unfranked dividend was valued at US$0.85/share and a final dividend unfranked of value US$1.00/share. Together they comprised of the total ordinary dividends of US$1.85/share, with US$4.24 earnings per share.
Also, an amount of US$832 million had been invested in R&D activities, of which 57% dedicated to novel product development, 13% for market development and 30% for lifecycle management. Total operating revenue of US$8,539 million and a Net Profit of US$1.9 billion has been reported for FY2019.
CSLâs Product Portfolio Performing Extraordinarily Well
CSL is involved in developing, marketing and delivering novel biotherapies as well as influenza vaccines via its two prime businesses CSL Behring and Seqirus.
CSLâs Largest Franchise, Immunoglobulin Recorded Remarkable Market Growth during 2019
CSL Behring Portfolio delivered a significant market growth reflected by a 16% hike in sales. It was notified to the market that CSLâs novel CDIP (Chronic Inflammatory Demyelinating Polyneuropathy) indication for HIZENTRA® and the inclusion of this CDIP indication for PRIVIGEN® played a remarkable role in driving CSLâs growth with â
- PRIVIGEN® sale shot up 23%
- HIZENTRA® sale up 22%
Furthermore, the augmented demand of immunoglobulin was driven by increased use of chronic therapies, improved awareness as well diagnosis of chronic diseases, CIDP indication as mentioned above & growing usage for SID. Likewise, there is a rising demand of patients for HAEGARDA® and KCENTRA®, which are CSLâs speciality products. Also, encouraging advancements in CSLâs Haemophilia portfolio contributed to its stupendous financial performance.
CSLâs other products that have been transformational regarding its solid growth include-
- ALBUMIN sales were up 15%
- IDELVION® sales grew by 40%. IDELVION® is CSLâs remedial treatment for Haemophilia B patients.
- HAEGARDA® sales raised by 61%. HAEGARDA® is the treatment for Hereditary Angioedema (HAE) patients.
- KCENTRA® sales rose 14%.
CSLâs Seqirus Portfolio Also Contributed to Strong Revenue Growth
It was reported that the total sales of this portfolio were up by 19%, and revenues were reported to be increased by 12%. The company is undergoing a transition in its portfolio into distinguished products.
Influenza vaccine sales grew by 19%
- The sales of FLAUD®, an indication of active immunisation in patients aged 65 years and older who are suffering from influenza, increased more than two folds.
- FLUAD® preferred recommendations in the United Kingdom and Australia.
- Delivery of future antigen capacity expansion was approved by Holly Springs â FCC 3.0.
- Convincing real-world effectiveness data for FLUCELVAX®, a cell-based influenza vaccine.
- In preventing influenza-like illness in the US 2017/18 season (where H3N3 was predominant) FLUCELVAX® Quadrivalent was found 36% more effective than the standard egg-based QIV. For the FLUCELVAX®, all strains were manufactured using the cell-specific seed for NH 2019/20 season.
- CSL had also undergone pandemic reservation agreements with Europe and Canada.
- Worldâs first adjuvanted, cell-based pandemic influenza vaccine, aH5N1c received FDA acceptance of submission.
CSL is looking forward to expanding the market for FLUCELVAX® by â
- Launching a NH 2019/20 in Europe.
- Dossier has already been submitted to TGA in Australia.
- CSL expects its launch in the private market in SH 2021.
There has been decent progress in fill & finish expansion process. The company anticipates that the Liverpool would become operational from SH 2021 and Holly Springs would become operational from NH 2022/23.
Rolled Out New Plasma Collection CentresÂ
- During FY2019, CSL delivered sustained growth in plasma collection network with 30 new centres being inaugurated the United States resulting in a total 221 centres in the US. In Germany, a total of 8 centres, total of 3 centres in Hungary and five new centres in China.
- To further expand its footprints, the company intends to open another 40 new centres in the next financial year (FY2020).
- CSL also purchased liquid saline & sodium citrate facility purchased in South Carolina US to support plasma collection.
Expected Net profit After Tax (NPAT) for FY2020
- It is expected that Seqirus would remain on the right track to deliver in line through prior guidance leveraging the benefits from product differentiation and process improvement.
- NPAT for the next year is expected between ~$2,050 million to $2,110 million on constant currency basis which is anticipated to grow ~7 - 10% on FY2019 NPAT.
- Revenue growth of ~6% expected to grow ~10% adj for GSP.
Latest ASX Announcement
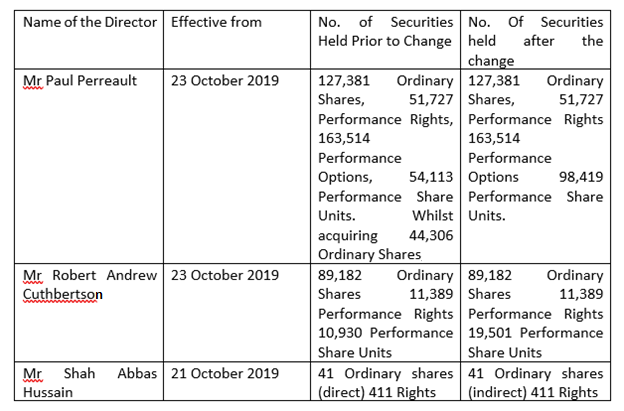
CSL released notice regarding the change of its directorsâ interest on 25 October 2019. It was informed that Mr Paul Perreault, Mr Robert Andrew Cuthbertson and Mr Shah Abbas Hussain changed their relevant interest in securities as per the section 205G of the Corporations Act. The details are given below.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.