Summary
- Several small-cap biotech and pharmaceutical companies have joined the battle against the coronavirus pandemic. They are contributing by either developing an effective drug/vaccine or diagnostic measures/tests to detect and curb the spread of the virus.
- Recce Pharmaceuticals compounds RECCE® 327 and RECCE® 529 will be tested against novel coronavirus by US researchers.
- The prevention and mitigation of SARS-CoV-2 infection for both the compounds will be assessed in an ex-vivo respiratory model system.
- AnteoTech Limited is developing a COVID-19 antigen rapid Point of Care (PoC) test.
The absence of a proven vaccine or treatment for COVID-19 has overshadowed the other challenges that we are currently facing relating the pandemic. With the ongoing surge in coronavirus cases, there is an urgent need to have advanced testing kits that can accurately detect the infection and at a quick pace.
COVID-19 has created havoc around the globe, and therapeutic interventions are urgently needed not only for curing severe COVID-19 cases but also for safeguarding the front-line health care service providers. As the process of drug development is complex and lengthy, several biotech companies have rushed to investigate and use drugs that have been authorised by regulatory agencies for other indications. The already approved drugs can be evaluated directly in late-stage studies as they do not need to conduct safety and tolerability trials again.
To prevent the rapid spread of COVID-19 worldwide, many pharmaceutical and biotech companies have joined the race either to develop a suitable drug/vaccine or to develop diagnostic measures to detect and curb the spread of the virus.
HAVE YOU READ: Lens on Global Healthcare Players Developing COVID-19 Treatment
With this in mind, we will acquaint you with two ASX-listed small-cap healthcare companies, Recce Pharmaceuticals and AnteoTech Limited, who are engaged in the fight against the pandemic and their recent updates.
US Researchers to Test RECCE® Compounds against SARS-CoV-2
Recce Pharmaceuticals Ltd (ASX:RCE) was founded in 2007 and is an emerging global leader for developing innovative broad-spectrum synthetic antibiotics. RECCE® 327 is a patented product of the Company developed for the treatment of blood infections and sepsis.
On 16 July 2020, Recce Pharmaceuticals disclosed that the Company had entered a partnership with Path BioAnalytics Inc (PBA), a US-based precision medicine company, for the study of RECCE® 327 and RECCE® 529 against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
As part of the collaboration, PBA Researchers will assess the activity of RECCE® 327 and RECCE® 529 against the novel coronavirus in an ex-vivo respiratory organoid model system.
The study will be conducted in the advanced Biosafety Level 3 containment labs of a US-based research university, where the infection of the organoids with novel coronavirus will take place.
Prevention/mitigation Assessment of RECCE® 327 and RECCE® 529
The study aims to assess the prevention/mitigation of both compound therapies for SARS-CoV-2 infections in an ex-vivo respiratory model system with initial data projected in September 2020. Notably, all intellectual properties (IPs) are retained by Recce Pharmaceuticals.
After the selection of a domestic program, as disclosed previously, the antiviral potential of compounds of Recce Pharma are being tested independently in Australia and the US.
Dr John Prendergast, Non-Executive Chairman, highlighted that in the past few months, Recce had received several expressions of interest from various universities as well as research organisations to collaborate and develop potential new treatments to address the unmet medical needs of COVID-19 patients.
Mr Prendergast added:

Stock Performance: On 17 July 2020, RCE stock declined by 4.138% to close the at A$1.390. With a market capitalisation of A$209.04 million, RCE has ~144.17 million shares trading on ASX. In the last three months, the stock has delivered an exceptional return of 281.58%.
AnteoTech Developing COVID-19 Test Platform
ASX-listed AnteoTech Limited (ASX:ADO) is engaged in Point of Care diagnostic testing and offers effective and efficient use of electricity through Li-ion batteries. The Company has IP rights in its core technology product groups AnteoReleaseTM, AnteoCoatTM, and AnteoBindTM.
On 16 July 2020, AnteoTech revealed that the Company had produced multiplexed proof of concept assays by applying an Axxin AX-2X-S Lateral Flow Reader designed to provide high sensitivity concurrent detection of COVID-19 and Flu A&B in a quick test format.
The rapid Point of Care (PoC) test for COVID-19 antigen intends to occupy a distinctive market position. Notably, the Company mentioned that the next phase of development, along with the clinical trial, will take six to nine months.
Outcomes of COVID-19 Test Development of AnteoTech-
The three tests- COVID-19 antigen, Flu A and Flu B harness the AnteoBind activated Europium particle technology of AnteoTech that is designed to enhance assay reproducibility and performance.
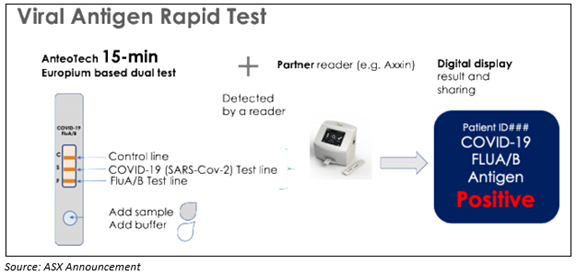
On completion, AnteoTech believes the test will full-fill an unmet medical need in the diagnostic response to COVID-19 by offering:
- Increased sensitivity and detection facilitated by the AnteoBind Europium particle conjugation. This allows earlier detection of the infection and the possibility for detection from mucus and saliva samples. As the Company holds IP for AnteoBind and has not licensed AnteoBind-activated Europium particles for COVID-19 antigen assays, no other COVID-19 test on the market has or will have AnteoBind-activated Europium as a basis for COVID-19 testing in the future.
- COVID-19 antigen test combined with Flu A&B in a multiplex platform for differentiating the cause of symptoms at the point of care.
Next phases aim to optimise the tests, improve further the lower limit of detection
The Proof of Concept for AnteoTech’s COVID-19 antigen test shows a working full cassette assay. The Company mentioned that the next phases of development will be conducted in the next 6-9 months.
The next phases will aim to optimise the tests, enhance further the lower limit of detection, verify & validate the design, conduct clinical trial required for targeted markets, obtain regulatory approvals and plan for outsourced scaled production.
Moreover, the regulatory registrations or approvals are required before market launch.
On completion, a portable point of care (PoC) device for swab samples is anticipated to offer precise identification within 15 minutes. The Company considers this capability will enable health care service providers to decrease the spread of COVID-19 via recommendation of immediate isolation of coronavirus infected people.
Notably, AnteoTech will reveal any partnership developments as they occur in the future.
Mr Derek Thomson, CEO AnteoTech commented:

Stock Performance: On 17 July 2020, ADO last quoted at A$0.004, a decline of 11.111% from its previous close. The market capitalisation stood at A$59.19 million with ~1.64 billion outstanding shares trading on the Australian Securities Exchange.







