What Causes Cancer?
Cancer is caused by the transformation of normal cells into tumour cells, which is a multistage process. The progress generally happens from a pre-cancerous lesion to a malignant tumour. An interaction between a personâs genetic factors and three categories of external agents lead to the above process. These three categories include physical carcinogens, such as ultraviolet and ionizing radiation; chemical carcinogens, such as asbestos, components of tobacco smoke, aflatoxin, and arsenic; and biological carcinogens, such as infections from certain viruses, bacteria, or parasites. Another factor that increases the risk of cancer development is ageing.
Key Statistics on Cancer
Being the 2nd leading cause of death around the world, cancer was responsible for approximately 9.6 Mn deaths in the year of 2018 as per stats submitted by the World Health Organization. The low and middle-income countries are most likely to be the victim of the deadly disease with the majority of deaths occurring in those regions. Lack of physical activity, use of tobacco and alcohol, low intake of healthy food and high body mass index are the few of the major reasons for around one-third of the deaths caused by cancer. Out of these, Tobacco is most likely to be the riskiest factor for cancer. The most common types of cancer include Lung, Breast, Colorectal, Prostate, Skin and Stomach cancer.
Early identification of cancer is the key to reduce the mortality rate. The diagnosis of cancer comprises three steps, which, when taken care of can help save many lives.
- Complete awareness about the disease among the population.
- Secondly, information on clinical evaluation, diagnosis and staging related to the disease.
- Third and the most important consideration is access to the treatment of cancer.
Kazia Therapeutics Limited (ASX:KZA)
Kazia Therapeutics Limited (ASX:KZA) develops pharmaceuticals drugs. It has a portfolio of development candidates diversified across several distinct technologies and conducts pharmaceutical research and development. The company produces innovative drugs for a range of cancers. In an announcement dated 20th May 2019, the company signed a collaboration agreement with the Alliance for Clinical Trials in Oncology Foundation to launch a multi-centre Phase II study to identify the potential use of Kaziaâs new drug, GDC-0084. An open-label Phase II study in patients with metastases will be conducted by Alliance. The trial will include up to 150 patients at multiple locations across the US. While Kazia will be responsible for providing support in the form of drug study and the financial grant, Alliance will take the lead in the study. The study is expected to begin recruitment in the second half of the calendar year 2019 and will take approximately two years to complete. According to another announcement dated 8th May 2019, The Bank of New York Mellon Corporation became a substantial shareholder of the company.
Financial Highlights
During the half-year ended 31 December 2018, the company reported a cash balance of $5.4 million as at 31st December 2018 when compared to $6 million as at 30th June 2018. The liquid assets amounted to $11.1 million in comparison to $12.5 million as at 30th June 2018. The reduction in liquid assets was largely due to a decline in the equity holding in Noxopharm Limited. During the period, the company paid $5.8 million for the advancement of R&D pipeline, which led to a notable reduction in cash burn from $9.2 million as on 31st December 2017. The company reported revenue from ordinary activities of $28,831, down 56.5% on the prior corresponding period value of $66,227. The operating expenses during the period amounted to $2,085,992 as compared to $4,456,589 in the prior corresponding period.
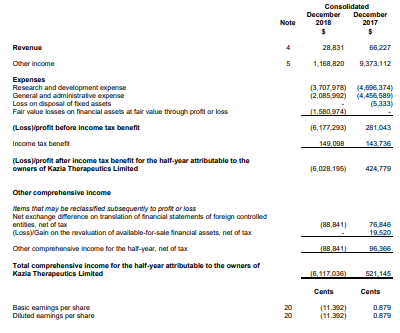
Statement of Profit & Loss for the period ended 31 December 2018 (Source: Company Reports)
The current period was adversely impacted by a non-cash fair value loss of $1,580,974 on shares and options held. The company incurred a loss of $6,028,195 during the current period, which went down on the prior corresponding period profit by 1519.1%.
Clinical Programs
The company has in place a lead program, GDC-0084, being developed in the most common and most aggressive form of primary brain cancer, glioblastoma. It is expecting to release an initial report on the Phase II study, which is performing well, during the first half of the calendar year 2019. The program in aggregate now comprises three active clinical studies, including the one mentioned above, Phase I trial in collaboration with St Jude Childrenâs Research Hospital focusing on a type of childhood brain cancer and another Phase II trial in collaboration with Dana-Farber Cancer Institute in breast cancer brain metastases. Kaziaâs Cantrixil program is also progressing well with a Phase I study in ovarian cancer underway. These two programs are critical in defining the future form and direction of the company.
Funding: The company saw huge participation in the Share Purchase Plan apart from the long-term institutional investors who joined the registry initially. Through the Share Purchase Plan, the company raised an amount of $0.8 million contributed by over 130 shareholders. This has increased the amount of total funds raised to $4.2 million.
Overall, the half-year period ending 31 December 2018 has not witnessed much of the company growth in terms of numbers, with the declining revenues and a lower cash balance in comparison to the prior corresponding period. The period also saw the operating expenses increase dramatically. Despite the above figures reported by the company, it has taken steps towards the development of the business with a lump sum amount spent on research and development for clinical trials of its products. With the research programs progressing well in the form of collaborations being undertaken by the company, there are chances that we will see Kazia shape a better future.
Stock Performance
The companyâs stock yielded returns of -19.59% and -29.09% over a period of one month and three months, respectively. The stock of the company is currently trading at $0.395, up 1.282% on 7th June 2019. The company holds a market capitalisation of $24.25 million.
Immutep Limited (ASX:IMM)
Immutep Limited (ASX:IMM) develops immunotherapy treatments for cancer and autoimmune diseases. The company engages in the treatment of cancer through the LAG3 immune control mechanism. Its key product is Eftilagimod Alpha, which is currently being tested in clinical trials to serve as a treatment for breast cancer and melanoma. In an announcement to the exchange dated 6th June 2019, the company notified the market about the enrolment of a very first patient for the INSIGHT-004 Phase I Clinical Trial. The patient has been enrolled in Germany. Being conducted in collaboration with Merck KGaA, Darmstadt, Germany and Pfizer Inc, the study is aimed at evaluating the safety and reliability of Eftilagimod Alpha, Immutepâs lead immunotherapy product.
On 3rd June 2019, the company announced the issue of 500,000 fully paid ordinary shares for exercise of Performance Rights under the Executive Incentive Plan into fully paid ordinary shares. The purpose underlying the issue was vesting and exercise of Performance Rights under the Executive Incentive Plan into fully paid ordinary shares. The European Patent Office granted IMM with another patent title âCombined Preparations for the Treatment of Cancerâ. The patent protects the intellectual property related to combined therapeutic preparations consisting of eftilagimood alpha, its lead active immunotherapy candidate.
As per the announcement for 1H19 earnings, the company reported no revenue as compared to $2.58 million in the prior corresponding period. The loss after tax for the period amounted to $8.67 million, up 100.7% on the prior corresponding period. However, the first half of FY19 was characterised by strong progress in its clinical development program and partnerships. The data reported from efti, the lead immunotherapy product, was favourable backed by the data from Phase I TACTI-mel clinical trial. It achieved a significant operational milestone during July 2018, after the approval of the companyâs investigational new drug application for efti by US Food and Drug Administration.
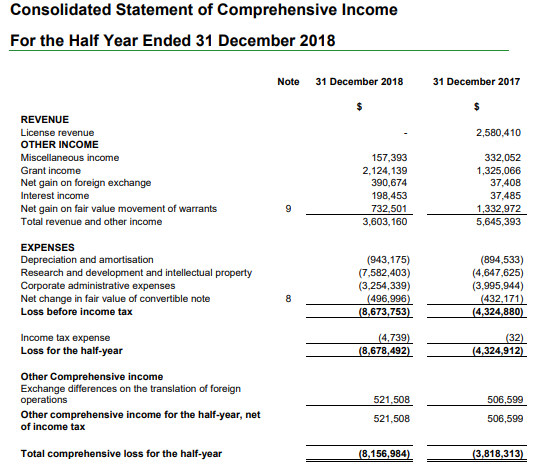
(Source: Company Reports)
To test eftilagimod alpha as part of a cancer peptide vaccine, the company entered into a clinical trial collaboration with CYTLIMIC in March 2019. The company received another patent, namely âthe derivatives thereof for Producing Monocyte Immune Responseâ or Use of Recombinant LAG-3â in the same month. For further business development, the company entered into another clinical trial collaboration with Merck KGaA, Darmstadt, Germany and Pfizer Inc. The agreement aimed at evaluating the combination of efti with avelumab. Immutep also completed US financing through NASDAQ listing and raised US$5.2 million in December 2018 to carry on with its LAG-3 clinical development programs.
Overall, IMMâs strategy of growth has been aligned with business development in the form of collaborations for clinical trials. The company has entered into partnerships with four of the worldâs best 10 pharmaceutical companies (as mentioned above).
The companyâs stock yielded return -22.86% over a period of three months. The stock of the company is currently trading at $0.026, down 3.704% during the dayâs trade with a market capitalisation of $91.49 million as on 7th June 2019.
Disclaimer
This website is a service of Kalkine Media Pty. Ltd. A.C.N. 629 651 672. The website has been prepared for informational purposes only and is not intended to be used as a complete source of information on any particular company. Kalkine Media does not in any way endorse or recommend individuals, products or services that may be discussed on this site. Our publications are NOT a solicitation or recommendation to buy, sell or hold. We are neither licensed nor qualified to provide investment advice.